1. INTRODUCTION
Okra, commonly known as “Bhindi” or “Lady’s finger” (Abelmoschus esculentus [L.] Moench), belonging to the Malvaceae family is an amphidiploid crop with chromosome number of 2n = 130. Being a warm-season vegetable, it is cultivated in tropical and subtropical parts of the world. It is a short duration annual, herbaceous, self-pollinated crop with monopodial growth habit, typically reaches heights of 1–2 m depending on the varieties and growing conditions [1]. The plant bears solitary, axillary flowers with a pentamerous structure, which are hermaphroditic in nature and exhibit a regular complete flower morphology. The type of inflorescence in okra is generally axillary solitary cymose, and the fruit is a capsule, commonly referred to as a pod, which is loculicidally dehiscent at maturity [2]. Nutritionally, okra is valued for its low caloric content and high dietary fiber, making it beneficial for weight management and blood sugar regulation. The pods are a rich source of Vitamin C, Vitamin K, folate, calcium, magnesium, and potassium, and contain a unique mucilaginous compound that supports digestive health and has medicinal applications. The antioxidant profile of okra includes flavonoids and polyphenols, which contribute to its role in preventing oxidative stress and related disorders [3]. Globally, India represents 60% (6.90 mt) of total world production (11.52 mt), followed by Nigeria 16% (1.90 mt), Mali 7% (0.76 mt), Pakistan 3% (0.31 mt), Sudan 3% (0.30 mt), Ivory Coast 2% (0.18 mt), Iraq and Bangladesh 1% (0.09 mt) (FAO, 2024) [4].
The evolving climatic conditions are occurring at a staggering rate and are impacting crop production, soil, water, and farm workers. Okra growth and production are affected by both abiotic (extreme temperatures, water scarcity, or waterlogging) and biotic stresses (diseases like YVMV). According to the United States Environmental Protection Agency (EPA), from early 2020, the Southwest U.S. has been facing one of the most intense and prolonged droughts in the past 1200 years [5]. According to the International Food Policy Research Institute (IFPRI) data, okra, maize, rice, and wheat production are adversely affected by water shortage [6]. Drought-induced yield losses can have adverse economic effects on farmers and the agricultural industry. Yield losses ranging from 30% to 100% have been documented in okra, mainly attributed to drought stress [7]. Approximately, a 21% decline in okra production and a 40% decline in maize production were recorded from 1980 to 2015 only because of water shortage [8].
Drought impacts seed germination and physio-biochemical functions, such as photosynthesis, mesophyll conductance, transpiration, and relative water content, which ultimately affects final yields of agronomic and horticultural crops [9]. Seed germination and seedling growth are crucial for plants to establish themselves, especially under stress. Drought stress in okra reveals its negative effect on plant growth by changing the plant biochemical content and negatively affecting photosynthesis. The studies show that there is a decrease in fresh weight, dry weight, plant height, chlorophyll content, carotenoids, and total protein, but proline and Membrane stability index (MSI) were highest under water stress conditions [10]. Drought-resistant plants employ various mechanisms to cope with water deficiency, such as osmotic adjustment, where they accumulate osmolytes, such as proline, sugars, and other solutes, to maintain cell turgor and protect cellular structures. Other key strategies include stomatal regulation to reduce water loss, deep or extensive root systems to access deeper water sources, enhanced water-use efficiency, and modifications in leaf morphology, such as smaller or wax-coated leaves, to minimize transpiration. In addition, these plants enhance their antioxidant defense system, producing higher levels of stress-induced antioxidant enzymes that help mitigate oxidative damage, which is critical for survival under drought [11]. Together, these adaptations enable plants to survive in arid environments. Identifying how okra responds to drought stress, farmers can adapt their practices and cultivars to maintain productivity during periods of water scarcity and reduce economic losses. More studies are needed to better understand the plant response toward water-deficit conditions.
In controlled conditions, polyethylene glycol (PEG) 6000 is commonly used in plant research to simulate drought stress due to its ability to induce osmotic stress without causing direct toxicity to plants [12]. The molecules of PEG are large and cannot penetrate the cell membranes, but lower the water potential of the solution by binding water molecules. This reduces free water availability around the root zone, effectively mimicking the conditions of drought by limiting water uptake [13]. While various studies have effectively simulated drought conditions by applying different concentrations of PEG [14], this study is the first to evaluate genotype-specific responses in okra under a wide gradient of PEG-induced water stress (0–30%). This study aims to differentiate genotypes based on their drought tolerance and systematically standardize an optimal concentration of PEG 6000 for stimulating drought conditions. By assessing root and shoot traits under different stress levels, this research provides a baseline for effective screening for drought resilience and selecting an appropriate PEG concentration for consistent simulation of drought conditions during early developmental stages.
2. MATERIALS AND METHODS
2.1. Experimental Site and Material
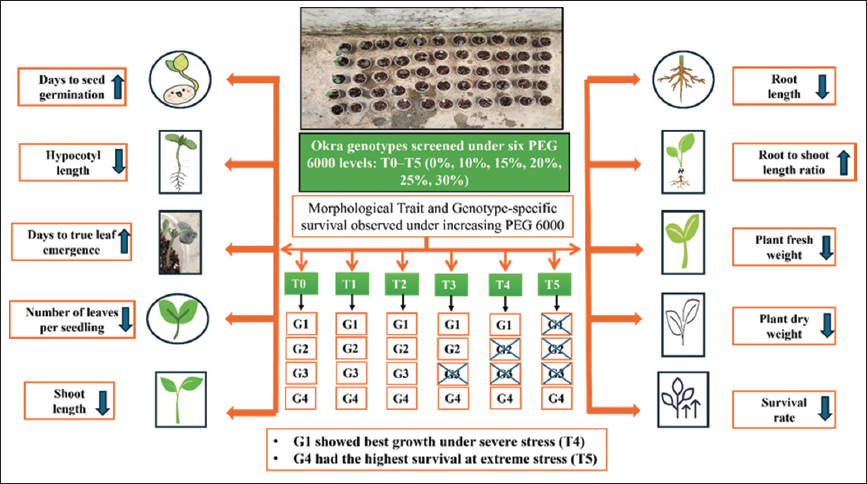
The present study was undertaken to standardize the optimal concentration of PEG 6000 for the induction of drought stress in okra under controlled climatic conditions. The experiment was carried out in a polyhouse, at Lovely Professional University, Phagwara, Punjab, India, during 2024. The research aimed to evaluate the impact of PEG-induced drought stress on the root and shoot development of four distinct okra varieties: Pusa Sawani, Bhindi Panchwati, MS-1031, and Sonam (referred to as G1, G2, G3, and G4, respectively). These varieties were tested to understand their differential responses at varying PEG levels of drought stress. The experimental set-up followed a completely randomized block design with three replications and six treatments.
2.2. Treatment Details and Methodology Adopted
To simulate drought conditions, PEG 6000, a widely used osmotic agent, was employed. Various concentrations of PEG 6000 were prepared by dissolving it in distilled water, following the protocol established by Michel and Kaufmann [15]. The concentration gradient was precisely chosen to cover a range of stress levels: T0 - control (distilled water), T1 - 10% PEG 6000, T2 - 15% PEG 6000, T3 - 20% PEG 6000, T4 - 25% PEG 6000, and T5 - 30% PEG 6000. This range was selected based on earlier studies demonstrating that 10–30% PEG 6000 effectively simulates mild to severe drought stress during seedling development in various crops, including maize and tomato [16]. The aim was to determine an optimal PEG level that imposes sufficient drought stress to influence plant growth while still allowing the plants to survive and exhibit measurable morphological responses.
At first, the seeds of four okra accessions were soaked overnight in water and then they were sown in plastic cups of 200 mL capacity containing cocopeat, vermicompost, and perlite. Six seeds in each cup were sown, which later were thinned to 2 and 15 mL of PEG 6000 from each concentration was added in the cups at a regular interval of 48 h starting from the date of sowing. The observations were recorded for ten traits, that is, Days to germination (DG), hypocotyl length (HL), days to true leaf emergence (DTLE), number of leaves per seedling (NLPS), shoot length (SL), root length (RL), root: Shoot length ratio (RSLR), survival rate (SR), plant fresh weight (PFW), and plant dry weight (PDW). To determine PFW and PDW, freshly harvested seedlings per replication were gently blotted with paper towels to remove excess surface moisture. Fresh weight was immediately measured using weighing balance (ATOM Selves- MH 200). The samples were then placed in a hot air oven (NSW India, Calton 143) at 80°C for 24 h until a constant weight was achieved and the dry weight was recorded using the same balance.
2.3. Statistical Analysis
Analysis of variance (ANOVA) was conducted using Minitab v 19.0 to evaluate the significance of variance attributed to genotypes, treatments, and their interaction (genotypes × treatments) for estimated traits in okra under water stress conditions. Descriptive statistics, clustering analysis, and Pearson’s correlation coefficients (r) for the estimated traits were also calculated using Minitab v 19.0 and R software [17].
3. RESULTS AND DISCUSSION
A standardization experiment was conducted using four okra genotypes (Pusa Sawani, Bhindi Panchwati, MS-1031, and Sonam) to identify the PEG 6000-induced stress levels suitable for effective drought tolerance screening. The results from ANOVA revealed highly significant differences (P < 0.01) among genotypes, treatments, and their interactions across all evaluated traits, including DG, HL, DTLE, and SL [Table 1]. These findings underscore the critical role of genotype selection in determining drought tolerance, as different genotypes show varying responses to PEG-induced stress. Treatments had a highly significant effect on all the traits, highlighting the effectiveness of PEG 6000 in creating varying levels of drought stress [Figure 1]. The significant interactions between genotypes and treatments suggest that the response to drought varies among okra cultivars, emphasizing the need for precise standardization of PEG6000 concentrations to optimize drought tolerance studies in these cultivars. In a similar study done in tomatoes, it also showed notable distinctions across all characteristics [18]. The results from Tukey pairwise comparisons test for almost all traits reveals that there are significant pairwise differences between treatment levels (T0-T5) under drought stress conditions [Figure 2]. Treatments at higher drought stress levels (T3-T5) showed a marked reduction in most growth traits, while lower stress levels (T0-T2) displayed less pronounced differences. As drought stress intensifies, plants’ ability to absorb and utilize water decreases, which causes their tolerance mechanisms to fail and hinders proper growth [19].
Table 1: Analysis of variance for okra traits under drought stress.
| Traits | Genotypes (df=3) | Treatments (df=5) | Genotypes*Treatments (df=15) | Error (df=46) |
|---|
| Days to germination | 50.00** | 71.01** | 46.63** | 6.81 |
| Hypocotyl length | 15.32** | 22.77** | 2.17** | 0.41 |
| Days to true leaf emergence | 300.80** | 277.69** | 119.63** | 18.4 |
| Number of leaves per seedling | 2.13** | 25.57** | 1.54** | 0.3 |
| Shoot length | 33.04** | 81.74** | 6.91** | 0.84 |
| Root length | 20.56** | 37.60** | 7.73** | 1.34 |
| Root-to-shoot length ratio | 1.62** | 0.81** | 0.34** | 0.09 |
| Plant fresh weight | 0.07** | 0.63** | 0.02** | 0 |
| Plant dry weight | 0.00** | 0.03** | 0.00** | 0 |
| Survival rate | 0.31** | 0.99** | 0.04** | 0 |
 | Figure 1: (a-d) Effect of polyethylene glycol 6000 concentrations on seedling growth of different okra varieties.
[Click here to view] |
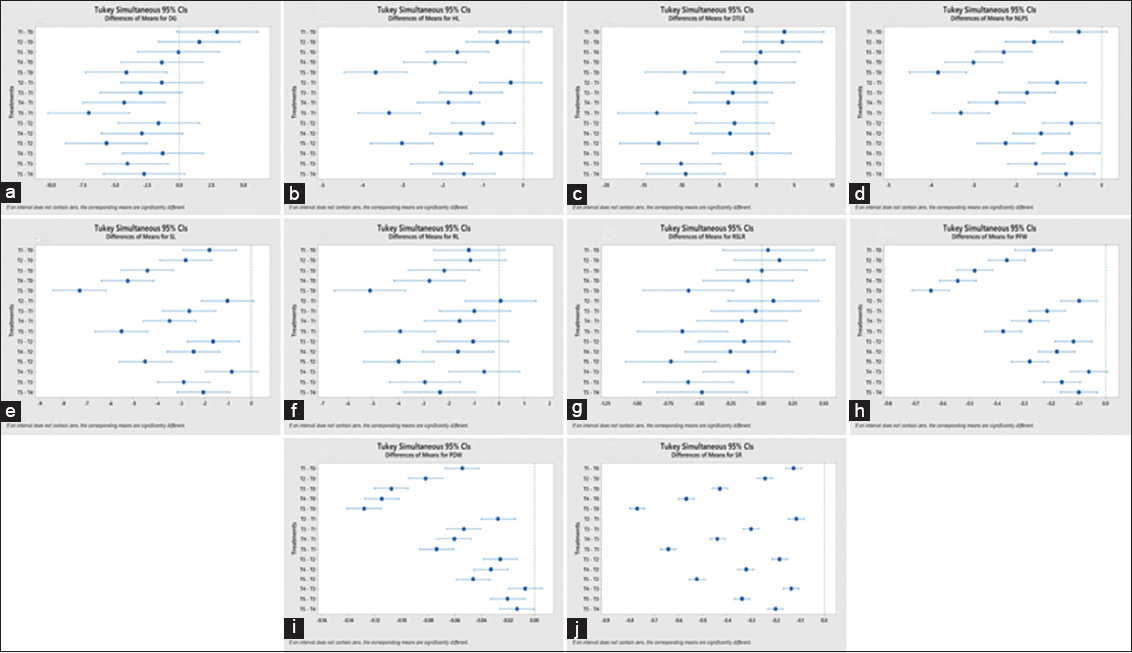 | Figure 2: (a-j) Tukey’s honestly significant difference pairwise comparisons of the effect of treatments.
[Click here to view] |
3.1. Descriptive Statistics
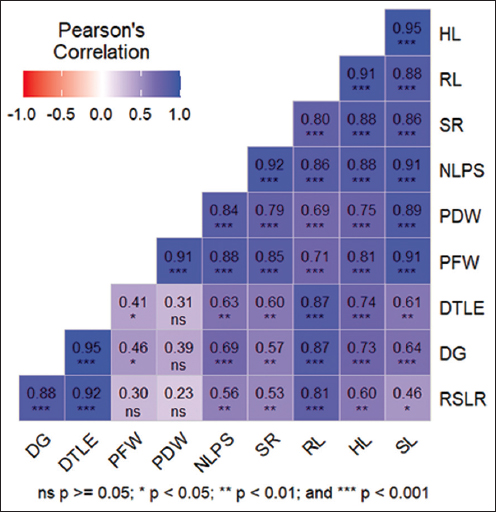
Descriptive statistical analysis was conducted for ten traits, as shown in Table 2. The mean values for these traits varied significantly, highlighting the variation in plant performance under the experimental conditions. DTLE recorded the highest mean value (11.27), indicating a longer duration for true leaf emergence, while PDW had the lowest mean value (0.048), and reflecting lower biomass accumulation under these conditions. Similarly, maize cultivars exposed to drought experienced reduced fresh and dry biomass compared to their controls, a result of the substantial drought-induced suppression of growth [20]. DTLE also had the highest standard deviation (8.35) and coefficient of variance (74.04%), which shows that there is significant variability between samples, while PDW had the lowest standard deviation (0.0487) but a high coefficient of variation (99.95%), indicating that there are significant differences in dry weight accumulation. The range of values from minimum to maximum highlights the variability in responses, DG ranges from 0 to 16 days, indicates significant variation in germination time, while SL, ranging from 0 to 10.1, reflects the sensitivity of shoot growth to different drought stress levels. A drought environment plays a critical role in seed germination percentages, significantly impacting the early growth of seedlings. The scarcity of water not only hinders the germination process but also poses a serious challenge to the development of young plants [21]. These variations suggest that drought stress has a substantial impact on both early germination and shoot development, with some plants showing delayed germination or reduced shoot growth under severe stress conditions.
Table 2: Descriptive statistics of okra traits under drought stress.
| Variable | Range | Mean±SE | Standard deviation | Coefficient of variation |
|---|
| DG | 0–16.0 | 6.43±0.55 | 4.63 | 71.92 |
| HL | 0–05.1 | 2.41±0.20 | 1.73 | 71.77 |
| DTLE | 0–28.0 | 11.27±0.98 | 8.35 | 74.04 |
| NLPS | 0–04.0 | 2.12±0.18 | 1.56 | 73.43 |
| SL | 0–10.1 | 3.94±0.36 | 3.03 | 76.92 |
| RL | 0–6.40 | 3.49±0.29 | 2.46 | 70.56 |
| RSLR | 0–1.93 | 0.66±0.05 | 0.51 | 76.45 |
| PFW | 0–0.76 | 0.26±0.02 | 0.23 | 86.56 |
| PDW | 0–0.18 | 0.04±0.00 | 0.04 | 99.95 |
| SR | 0–0.88 | 0.43±0.03 | 0.30 | 70.19 |
3.2. Impact of PEG on Genotype Morphology
3.2.1. Pusa Sawani
Under increasing concentrations from T0 to T4 [Figure 3a and b], Pusa Sawani exhibited delayed DG and DTLE, accompanied by reduced HL, SL, and RL. SR decreased significantly up to T4 whereas no plant was able to survive at 30% (T5). Both PFW and PDW also decreased significantly indicating adverse effects of water stress on growth and plant viability. The increased RSLR indicates that shoot growth was more impacted compared to RL under drought conditions [Table 3]. Consistent with previous studies, water stress not only reduced dry matter accumulation in both the shoots and roots but also led to a greater reduction in the root system, ultimately leading to an increased root-to-shoot ratio [22].
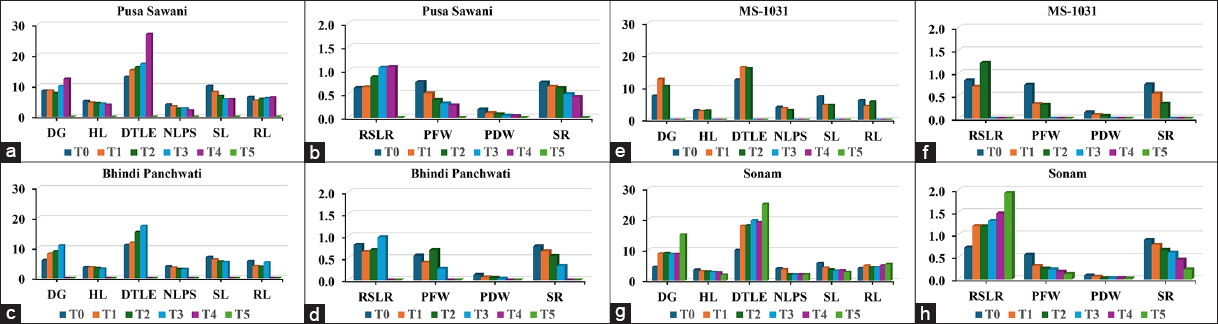 | Figure 3: (a-h) Effect of polyethylene glycol 6000 concentrations on morphological traits of genotypes under drought stress conditions.
[Click here to view] |
Table 3: Effect of polyethylene glycol-induced drought stress on morphological traits of okra genotypes across different treatment levels.
| Genotypes | Treatments | DG | HL | DTLE | NLPS | SL | RL | RSLR | PFW | PDW | SR (%) |
|---|
| Pusa Sawani | T0 | 8.50 | 5.1 | 13.00 | 4.00 | 10.1 | 6.4 | 0.63 | 0.76 | 0.18 | 75 |
| Pusa Sawani | T1 | 8.50 | 4.6 | 15.17 | 3.33 | 8.05 | 5.22 | 0.65 | 0.52 | 0.10 | 66 |
| Pusa Sawani | T2 | 7.67 | 4.48 | 16.17 | 2.67 | 6.73 | 5.82 | 0.86 | 0.38 | 0.08 | 63 |
| Pusa Sawani | T3 | 10.00 | 4.3 | 17.25 | 2.75 | 5.68 | 6.00 | 1.07 | 0.31 | 0.05 | 50 |
| Pusa Sawani | T4 | 12.33 | 3.9 | 27.00 | 2.00 | 5.73 | 6.20 | 1.08 | 0.26 | 0.03 | 44% |
| Pusa Sawani | T5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0% |
| Bhindi Panchwati | T0 | 6.00 | 3.65 | 11.00 | 4.00 | 7.10 | 5.70 | 0.80 | 0.56 | 0.12 | 77% |
| Bhindi Panchwati | T1 | 8.17 | 3.60 | 11.83 | 3.50 | 6.18 | 4.03 | 0.65 | 0.40 | 0.07 | 66% |
| Bhindi Panchwati | T2 | 9.00 | 3.37 | 15.33 | 3.00 | 5.55 | 3.82 | 0.69 | 0.69 | 0.05 | 55% |
| Bhindi Panchwati | T3 | 10.83 | 3.10 | 17.33 | 3.00 | 5.37 | 5.23 | 0.98 | 0.26 | 0.03 | 33% |
| Bhindi Panchwati | T4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0% |
| Bhindi Panchwati | T5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0% |
| MS-1031 | T0 | 7.50 | 2.95 | 12.50 | 4.00 | 7.25 | 6.10 | 0.84 | 0.74 | 0.13 | 75 |
| MS-1031 | T1 | 12.67 | 2.70 | 16.33 | 3.67 | 4.60 | 4.27 | 0.70 | 0.32 | 0.08 | 55 |
| MS-1031 | T2 | 10.50 | 2.85 | 16.00 | 3.00 | 4.60 | 5.65 | 1.23 | 0.31 | 0.06 | 33 |
| MS-1031 | T3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| MS-1031 | T4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| MS-1031 | T5 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Sonam | T0 | 4.50 | 3.60 | 10.00 | 4.00 | 5.70 | 4.05 | 0.71 | 0.55 | 0.08 | 88 |
| Sonam | T1 | 8.83 | 3.07 | 17.83 | 3.67 | 4.20 | 4.98 | 1.19 | 0.30 | 0.06 | 77 |
| Sonam | T2 | 9.00 | 2.97 | 18.00 | 2.00 | 3.63 | 4.30 | 1.18 | 0.24 | 0.02 | 66 |
| Sonam | T3 | 8.67 | 2.78 | 19.67 | 2.00 | 3.32 | 4.33 | 1.31 | 0.22 | 0.03 | 60 |
| Sonam | T4 | 8.67 | 2.63 | 19.00 | 2.00 | 3.40 | 5.00 | 1.47 | 0.17 | 0.03 | 44 |
| Sonam | T5 | 15.00 | 1.90 | 25.00 | 2.00 | 2.80 | 5.40 | 1.93 | 0.12 | 0.02 | 22 |
3.2.2. Bhindi panchwati
Bhindi Panchwati, when exposed to increasing levels of drought stress, leads to delayed DG and DTLE, along with a reduction in HL, NLPS, SL, and RL [Figure 3c and d]. In a similar study on canola, drought stress was found to increase the average number of DG and highlighted the varied responses of different species to water stress [23]. Plants exposed to higher concentrations of 25% (T4) and 30% (T5) were not able to survive and there was an observable decrease in PFW and PDW. The RSLR peaked at 20% (T3) which indicates greater resilience in root growth under moderate drought conditions.
3.2.3. MS-1031
MS-1031, when subjected to increasing concentrations of PEG 6000, showed delayed germination and true leaf emergence, along with reductions in HL, NLPS, SL, and RL [Figure 3e and f]. In line with these findings, Kumar et al. observed that as PEG concentration increased, key growth parameters, such as germination percentage, germination rate, root and SLs, and both root and dry weights consistently declined during seedling development [24]. Plants exposed to higher concentrations of 20% (T3), 25% (T4), and 30% (T5) did not survive, and significant reductions in both fresh and dry weights were observed. The RSLR was highest at 15% (T2), indicating stronger root growth under moderate drought stress [Table 3].
3.2.4. Sonam
Sonam under higher water stress conditions showed delayed germination and DTLE, accompanied by reduced HL, NLPS, SL, and both PFW and PDW [Table 3]. These responses are driven by lower water content, decreased turgor pressure, and reduced water potential which ultimately leads to low fresh and dry weight as well as reduced growth [25]. However, there was a slight increase in RL at 25% (T4) and 30% (T5), while the SR steadily declined as stress levels increased [Figure 3g and h].
REFERENCES
1. Massrie KD. Constraints and opportunities on okra (Abelmoschus esculentus) production in Ethiopia:A review. Front Sustain Food Syst. 2025;9:1546995. [CrossRef]
2. Kiran SC, Deshmukh HK, Harne SS, Taide YB, Komal VK, Nandanwar PS, et al. Diversity of bee flora in Dr. Panjabrao Deshmukh Krishi Vidyapeeth, Akola, Maharashtra, India. Indian J Ecol. 2023;50(3):725-36. [CrossRef]
3. Gemede HF, Ratta N, Haki GD, Woldegiorgis AZ, Beyene F. Nutritional quality and health benefits of okra (Abelmoschus esculentus): A review. Int J Nutr Food Sci. 2015;25:16-25. [CrossRef]
4. Statistical Report. Rome: Food and Agriculture Organization (FAO). International Crop Production Statistics;2024.
5. Statistical Report. United States Environmental Protection Agency (EPA). Southwest U.S. Drought Analysis;2020.
6. Statistical Report. International Food Policy Research Institute (IFPRI). Impact of Water Shortage on Crop Production;2023.
7. Mkhabela SS, Shimelis H, Gerrano AS, Mashilo J. Drought tolerance assessment of okra (Abelmoschus esculentus [L.] moench) accessions based on leaf gas exchange and chlorophyll fluorescence. Life (Basel). 2023;13(3):682. [CrossRef]
8. Siddique S, Naveed M, Yaseen M, Shahbaz M. Exploring potential of seed endophytic bacteria for enhancing drought stress resilience in maize (Zea mays L.). Sustainability. 2022;14(2):673. [CrossRef]
9. Jabborova D, Kannepalli A, Davranov K, Narimanov A, Enakiev Y, Syed A, et al. Co-inoculation of rhizobacteria promotes growth, yield, and nutrient contents in soybean and improves soil enzymes and nutrients under drought conditions. Sci Rep. 2021;11(1):22081. [CrossRef]
10. Ayub Q, Hussain I, Naveed K, Ali S, Mehmood A, Khan MJ, et al. Responses of different okra (Abelmoschus esculentus) cultivars to water deficit conditions. J Hortic Sci. 2021;16(1):53-63. [CrossRef]
11. Kusvuran S. Influence of drought stress on growth, ion accumulation and antioxidative enzymes in okra genotypes. Int J Agric Biol. 2012;14(3):401-406.
12. Castañeda V, González EM. Strategies to apply water-deficit stress: Similarities and disparities at the whole plant metabolism level in Medicago truncatula. Int J Mol Sci. 2021;22:2813. [CrossRef]
13. Ahmad MA, Javed R, Adeel M, Rizwan M, Yang Y. PEG 6000-stimulated drought stress improves the attributes of in vitro growth, steviol glycosides production, and antioxidant activities in Stevia rebaudiana Bertoni. Plants (Basel). 2020;9(11):1552. [CrossRef]
14. Hernández-Pérez CA, Gómez-Merino FC, Spinoso-Castillo JL, Bello-Bello JJ. In vitro screening of sugarcane cultivars (Saccharum spp. hybrids) for tolerance to polyethylene glycol-induced water stress. Agronomy. 2021;11(3):598. [CrossRef]
15. Michel BE, Kaufmann MR. The osmotic potential of polyethylene glycol 6000. Plant Physiol. 1973;51(5):914-6. [CrossRef]
16. Ul Sajjad Z, Fatima Z, Awais M, Waheed A, Ali A, Hashmi QA. Effect of polyethylene glycol (PEG) on germination and seedling parameters of maize varieties. Asian J Res Crop Sci. 2023;8(4):410-20. [CrossRef]
17. RStudio Team (2022). RStudio: Integrated Development for R.RStudio, PBC, Boston, MA, USA. Available online at: http://www.rstudio.com/
18. Akram S, Saleem Y, Khan AR, Wadood A, Hameed A, Sajjad S. Identification of drought-tolerant tomato genotypes using multi-trait index at early growth stage. J Soil Sci Plant Nutr. 2024;24:1-3. [CrossRef]
19. Islam MM, Kayesh E, Zaman E, Urmi TA, Haque MM. Evaluation of rice (Oryza sativa L.) genotypes for drought tolerance at germination and early seedling stage. Agriculturists. 2018;16:44. [CrossRef]
20. Efeo?lu B, Ekmekçi Y, Çiçek NU. Physiological responses of three maize cultivars to drought stress and recovery. S Afr J Bot. 2009;75(1):34-42. [CrossRef]
21. Li H, Li X, Zhang D, Liu H, Guan K. Effects of drought stress on the seed germination and early seedling growth of the endemic desert plant Eremosparton songoricum (Fabaceae). EXCLI J. 2013;12:89-101.
22. Kou X, Han W, Kang J. Responses of root system architecture to water stress at multiple levels:A meta-analysis of trials under controlled conditions. Front Plant Sci. 2022;13:10?9. [CrossRef]
23. Rezayian M, Niknam V, Ebrahimzadeh H. Effects of drought stress on the seedling growth, development, and metabolic activity in different cultivars of canola. Soil Sci Plant Nutr. 2018;64(3):360-9. [CrossRef]
24. Kumar PA, Reddy NN, Lakshmi NJ. PEG induced screening for drought tolerance in tomato genotypes. Int J Curr Microbiol Appl Sci. 2017;6(7):168-81. [CrossRef]
25. Guo YY, Yu HY, Yang MM, Kong DS, Zhang YJ. Effect of drought stress on lipid peroxidation, osmotic adjustment and antioxidant enzyme activity of leaves and roots of Lycium ruthenicum Murr. seedling. Russian J Plant Physiol. 2018;65:244-50. [CrossRef]
26. Zhang J, Mason AS, Wu J, Liu S, Zhang X, Luo T, et al. Identification of putative candidate genes for water stress tolerance in canola (Brassica napus). Front Plant Sci. 2015;6:1058. [CrossRef]
27. Bukan M, Kereša S, Peji?I, Sudari?A, Lovri?A, Šar?evi?H. Variability of root and shoot traits under PEG-induced drought stress at an early vegetative growth stage of soybean. Agronomy. 2024;14(6):1188. [CrossRef]
28. Islam MR, Hossain A, Akhter MM, Alam MA, Azam MG, Hossain J, et al. Evaluation of Polyethylene glycol (PEG-6000) induced drought stress tolerant Mungbean genotypes by using correlation, principal component, hierarchical clustering and multi-trait genotype-ideotypes distance index analysis. Curr Appl Sci Technol. 2023;25:e0260871. [CrossRef]
29. Cui G, Zhao Y, Zhang J, Chao M, Xie K, Zhang C, et al. Proteomic analysis of the similarities and differences of soil drought and polyethylene glycol stress responses in wheat (Triticum aestivum L.). Plant Mol Biol. 2019;100(4):391-410. [CrossRef]