1. INTRODUCTION
Tomato (Solanum lycopersicum L.) is an economically important agricultural commodity. It provides several nutrients including lycopene, beta carotene, flavonoids, Vitamin C, hydroxycinnamic derivatives and antioxidants that reduce risks in cancer, cardiovascular diseases, and Vitamin A deficiency [1]. In 2019, tomato was cultivated in a total of 16,360 ha of land yielding 223 thousand tons of fruit harvest, in the Philippines [2].
Due to increasing demand, tomato has been the subject of research focusing on improving fruit quality and developing disease resistance and stress tolerance through genetic transformation and other new breeding techniques. However, success in transformation relies on establishing an efficient regeneration system. Although there was significant progress in tomato transformation [3], successful transformation was confined to model cultivars such as Moneymaker [4], Micro-Tom [5,6], and Rio Grande [7].
Plant regeneration can be affected by several factors, including plant’s genetic makeup, type of explant, components of the culture medium, concentration, and type of plant growth regulators, and their interaction [8]. In tomato, plant growth regulators specifically auxin and cytokinin source and concentration, play a significant role in mediating the morphogenic capacity of the explant [9]. The addition of 2.0 mg/L kinetin and 0.5 mg-1 indole acetic acid (IAA) on Chu (N6) medium was found to be effective in enhancing plant regeneration percentage and number of shoots in tomato [10]. High frequency of shoot regeneration was also observed on Murashige and Skoog (MS) medium supplemented with 0.1 mg-1 IAA, 1.0 mg-1 Zeatin and 2.0 mg-1 benzylamino purine (BAP) along with 8–10 mg-1 silver nitrate in tomato varieties Rio Grande, Roma, and Moneymaker [11]. Adventitious shoot regeneration from the hypocotyl and cotyledon explants of Micro-Tom tomato was observed on MS medium containing 1.0 mg-1 Zeatin and 0.1 mg/L IAA [12].
The recalcitrance of tomato in in vitro culture makes it difficult to standardize one protocol for plant regeneration that will work for various genotypes, particularly in identifying plant growth hormones. With the advent of synthetic plant growth hormones, overcoming the phenomenon of in vitro recalcitrance is made possible. Thidiazuron (TDZ) is a synthetic cytokinin that is highly active even at lower concentration and is more resistant to the degrading enzymes in plants [13]. Due to its high activity, exposing an explant to TDZ can trigger various morphogenetic responses and will still be effective even after transferring to a medium devoid of TDZ, especially when inducing in vitro regeneration [14]. Specifically, it is known for its high potency for in vitro morphogenesis responses such as shoot proliferation and regeneration [15]. Its supplementation in the media applied whether at low or high concentrations can effectively induce in vitro shoot responses as observed in several crops such as date palm, bamboo, and apple [16-18].
An efficient regeneration system for tomato genotype Vb-15 is vital for its further genetic manipulation. This genotype is originally a Florida line (FLA-456) bred from Solanum chilense LA2779 and Royal Sluis tomato hybrid “Tyking” [19]. It has resistance to several begomoviruses, including the Tomato yellow leaf curl virus and has a high intensity of orange fruit color. Silencing of phytoene desaturase (PDS) gene involved in carotenoid biosynthesis in tomato was previously described in cv. Micro-Tom. This causes photobleaching mainly on fruit and leaves expressing that PDS gene has been knocked out [20]. Similarly, a clear manifestation of color change can be observed in Vb-15 to validate lycopene cyclase gene knockout using the Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) and associated protein-9 (CRISPR/Cas9) vector system delivered by Agrobacterium tumefaciens. However, preliminary regeneration experiments showed that Vb-15 is recalcitrant in in vitro culture.
This study aimed to develop an efficient regeneration system for tomato genotype Vb-15. The objectives were to select the best medium for callus induction and shoot regeneration and determine differences in morphogenic potential of the cotyledon explant in the presence of TDZ, BAP, and naphthalene acetic acid (NAA). The study also aimed to evaluate the reproducibility of the best medium on other tomato genotypes and to assess if the regeneration protocol developed can be utilized as a general procedure for future transformation studies.
2. MATERIALS AND METHODS
2.1. Preparation of Plant Materials
Seeds of tomato genotypes used in the study were obtained from the collections of Institute of Plant Breeding - University of the Philippines Los Baños (14.1546° N, 121.2631° E). The tomato variety Vb-15 was obtained from the Genetics Laboratory, tomato hybrids (Apollo and Improved Apollo) from Plant Physiology Laboratory, open-pollinated varieties (Discovery, Rosanna, and Northern Red) from the National Seed Foundation; and open-pollinated tomato breeding lines (Tm 14-1-1-5, Tm 15-26-0-10, Tm 163147, and Tm 12-18-0-8) from the Vegetables Section. Seeds were surface disinfected with 10% sodium hypochlorite (NaOCl, 5.25% a.i.) for 30 min and rinsed with sterile distilled water. These were soaked overnight followed by the same disinfection procedure before inoculating on in vitro germination medium (refer to 2.4).
2.2. Selection of Best Plant Growth Hormones for Callus Induction and Shoot Regeneration
Cotyledon explants were excised from the in vitro germinated seeds after 10–12 days. The explants were cut in half with approximately 0.5 cm in length and cultured on MS medium supplemented with BAP, TDZ, and NAA treatments with the abaxial side down.
2.3. Assessment of Shoot Elongation, Rooting and Greenhouse Establishment after Prolonged Cytokinin Treatment
Regenerated shoots through direct and from callus were excised after 12 weeks and were then transferred to MS basal medium for shoot elongation and rooting. After 1 week of acclimatization, well-rooted plantlets were cultured in sterilize soil mixture (garden soil: coir dust, 1:1, v/v) and maintained in the greenhouse until flowering.
2.4. Culture Condition and Maintenance
The culture medium for all experiments was based on Murashige and Skoog [21] salts and vitamins with 3% sucrose and solidified with 0.8% plant agar. The in vitro germination medium contained MS basal medium alone. For the selection of best plant growth hormones, the following were added (in mgl-1: T1- 0.5 BAP, T2 - 2.5 BAP, T3 - 0.5 TDZ, T4 - 2.5 TDZ, T5 - 0.5 NAA, T6 - 2.5 NAA, T7 - 0.5 BAP + 0.5 TDZ, T8 - 2.5 BAP + 0.5 TDZ, T9 - 0.5 BAP + 2.5 TDZ, T10 - 2.5 BAP + 2.5 TDZ, T11 - 0.5 BAP + 0.5 TDZ + 0.5 NAA, T12 - 2.5 BAP + 0.5 TDZ + 0.5 NAA. Moreover, the medium used to screen the in vitro response of ten tomato genotypes was MS supplemented with 0.5 mg-1 TDZ. The culture medium was adjusted to pH 5.6 and autoclaved at 15 psi for 20 min. Cultures were transferred to fresh medium with the same composition as the initial medium for two subculture cycles and were transferred to MS basal medium for shoot elongation and rooting. All cultures were maintained under 16-h photoperiod using cool white light emitting diode emitting a light intensity of approximately 30–50 μmol/(m2·s).
2.5. Evaluation of the In Vitro Response and Statistical Analysis
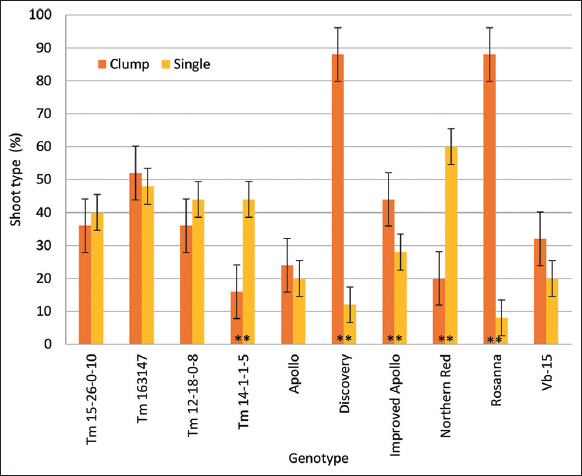
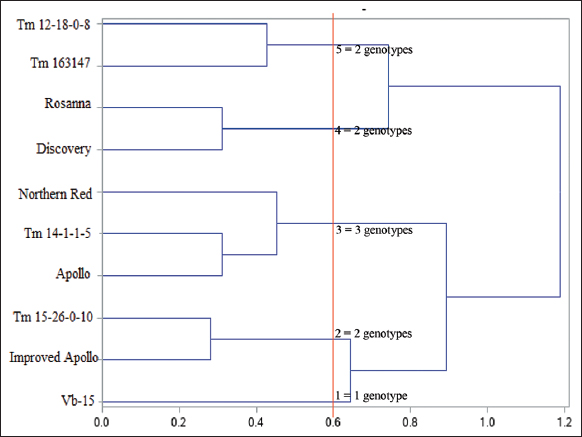
A total of 252 cotyledons with seven replicates having three samples were studied for the 12 treatments tested in Complete Randomized Design with subsampling. In callus induction and regeneration experiments, percent callus formation, callusing index (CI), percent shoot formation, and number of shoots were collected after 4 weeks of incubation. For the CI, the scale was: 0 - no tissue growth to swelling of the cotyledon with no callus formation; 1 - callus formation on one cut end of the cotyledon; 2 - callus formation on both cut ends of the cotyledon; and 3 - calli growth has completely covered the cotyledon explant and with at least two-fold increase in mass. For the experiment on the assessment of the reproducibility of the optimized treatment combination, 25 cotyledons per genotype with five replications were studied and laid out in a Complete Randomized Design. In vitro response diversity on the ten genotypes was analyzed using hierarchical cluster analysis based on Euclidean distance matrix. In addition, shoot characteristics were also classified into single – those with complete shoot structure and distinct apical meristem; clump – those with leafy shoot structure without distinct meristem. For all experiments, differences on the mean values were assessed at 95% confidence level (P ≤ 0.05). Means for each treatment and the standard deviation were calculated. Data analysis was performed using the analysis of variance and Least Significant Difference Test for CI and number of regenerants. Regeneration efficiency of responsive explants, callus, and shoot were obtained by quantifying the responsive cultures for each stage, and a binary logistic regression analysis was performed to analyze the data obtained. A student t-test was also performed to compare means per genotype. All analysis was processed in Statistical Analysis Software (version 3.8).
3. RESULTS
3.1. Callus Induction
The Vb-15 tomato seeds germinated with a relatively high efficiency (>85%). Cotyledon segments were obtained when they reached approximately 1 cm in length. During the first 5 days of incubation on the modified media, cotyledons started to swell followed by formation of cell clusters at the margin. Callus induction was effective in all treatments except for medium containing NAA alone which only produced adventitious roots. However, combining BAP, TDZ, and NAA at relatively low concentration induced rapid callus proliferation.
The ability of plant growth hormones in inducing callus growth was measured using CI where a significant variation (P = 0.0232) was observed. Maximum response was attained in MS + 0.5 mg-1 BAP + 0.5 TDZ mg-1 + 0.5 mg-1 NAA, with the highest CI of 2.94 [Table 1]. Although, there was a similar efficiency when BAP alone, TDZ alone, and their combination was used, explants cultured on MS + 0.5 mg-1 BAP + 0.5 mg-1 TDZ + 0.5 mg-1 NAA have more callus graded as 3 (mean CI difference = 1.26) compared to MS + 0.5 mg-1 BAP + 0.5 mg-1 TDZ. This only showed that adding NAA promotes rapid callus proliferation.
Table 1: Quantitative growth response of Vb-15 relative to different concentrations and combination of plant growth regulators.
| Treatment (in mgl-1) | % RE | % Callus formation per RE | Mean callusing index | % Shoot formation per RE | No. of regenerated shoot per RE | Highest shoot length (cm)** |
|---|---|---|---|---|---|---|
| 0.5 BAP | 94.44 | 100 | 1.94±1.01ab | 41.18a | 2.45±0.59b | 3.91±0.83a |
| 2.5 BAP | 100 | 100 | 2.61±0.50ab | 27.78a | 2.18±0.55b | 2.64±0.78b |
| 0.5 TDZ | 90 | 100 | 1.90±0.85ab | 55.6a | 4.45±0.92a | 3.22±0.35ab |
| 2.5 TDZ | 100 | 100 | 2.45±0.76ab | 55a | 4.64±0.68a | 1.74±0.42c |
| 0.5 NAA | 0 | 0 | 0c | 0a | 0c | NA |
| 2.5 NAA | 27.78 | 40 | 0.11±0.32c | 0a | 0c | NA |
| 0.5 BAP+0.5 TDZ | 89.47 | 94.12 | 1.68±1.11b | 35.29a | 2.27±0.54b | 1.17±0.15d |
| 2.5 BAP+0.5 TDZ | 89.47 | 100 | 2.16±1.01ab | 35.29a | 2.91±0.78ab | 1.60±0.15c |
| 0.5 BAP+2.5 TDZ | 89.47 | 100 | 2.11±0.88ab | 41.18a | 3.00±0.57ab | 1.61±0.13c |
| 2.5 BAP+2.5 TDZ | 94.44 | 100 | 2.33±0.77ab | 58.82a | 2.63±0.39b | 0.95±0.07e |
| 0.5 BAP+0.5 TDZ+0.5 NAA | 88.89 | 100 | 2.94±1.16a | 0a | 0c | NA |
| 2.5 BAP+0.5 TDZ+0.5 NAA | 94.44 | 100 | 2.33±0.77ab | 0a | 0c | NA |
*Means±the standard error with the same letter are not significantly different using least significant difference.
Measured from regenerated plantlets transferred to MS basal medium after 4 weeks, RE: Responsive explants.
3.2. Shoot Regeneration
Shoot differentiation started when the callus showed protruding clusters of green structures [Figure 1a] which later developed into torpedo structure [Figure 1b] and eventually showed early cotyledonary stage of shoots [Figure 1c]. On the other hand, some cultures induced adventitious shoot formation from the cotyledon explant under TDZ-treated medium during the early incubation period [Figure 1d]. These structures were stunted and developed large leafy structures. Regeneration was only observed in cultures inoculated on a medium devoid of NAA, showing that shoot formation was highly cytokinin-dependent [Table 1].
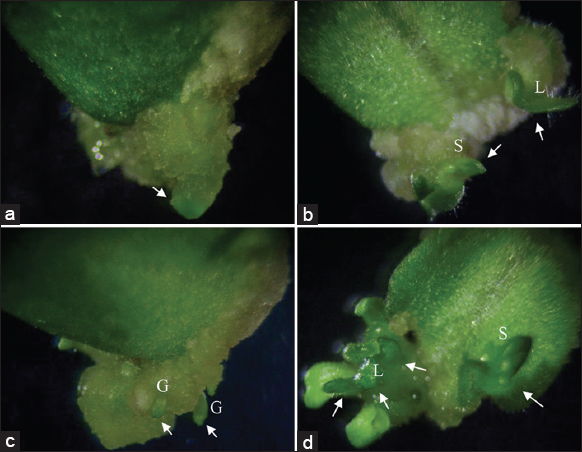 | Figure 1: Different morphological structures observed after 17 days of culture from the cotyledon explants of Vb-15. (a) Callus formation and green color of friable callus, (b) dedifferentiation of callus into leaf (L) and shoot (S) structures, (c) formation of globular (G) structures from friable callus and (d) direct shoot regeneration (S) and leaf organogenesis (L). Arrow indicates structures which developed into shoots at different stages from callus initiation to shoot formation. [Click here to view] |
This study demonstrated the significant role of cytokinin on shoot formation. Based on the results, there is a progressive increase in percent shoot formation in the presence of TDZ or when combined with BAP [Table 1]. The highest shoot percentage (58.82%) was obtained in high concentrations of TDZ and BAP both at 2.5 mg-1. In addition, the regeneration frequency, measured by counting the number of shoots formed per callus and shoot formation, was highly dependent on the concentration and type of plant growth hormones present in the media (P = 0.0439). The highest number of regenerated shoots was observed in MS medium fortified with 2.5 mg-1 TDZ with 4.64 shoots per callus. Although it has a similar efficacy (P = 0.8428), the use of 0.5 mg-1 TDZ has a higher percentage of shoot formation (55.6%). Regenerants from this treatment have distinct apical meristem and can grow into a complete shoot structure as compared with regenerants from 2.5 mg-1 TDZ that has a leafy structure of blinded shoots (without distinct meristem) and are usually stunted. A significantly higher frequency of shoots was obtained on TDZ medium than on BAP medium. Statistical analysis showed that explants cultured on TDZ would yield approximately two to three more shoots than on BAP. However, a combination of BAP and TDZ in the medium reduced the number of regenerated shoots manifesting the negative effect of BAP in multiple shoot organogenesis.
3.3. Shoot Elongation, Rooting, and Potting Out
All regenerated shoots were transferred to MS basal medium for shoot elongation and rooting [Figure 2]. After 4 weeks of incubation, maximum height in cultures was obtained on shoots previously treated with 0.5 mg-1 BAP and 0.5 mg-1 TDZ [Table 1]. However, regenerants from other treatments continue to produce multiple shoots showing that apical dominance was inhibited. Further incubation on the basal medium induced root formation (100% of the cultures). Before transferring to sterilized soil mixture, the plantlets were acclimatized for 1 week when they reached approximately 5 cm in height. Relatively high survival rate (75–78%) was obtained on well-developed plantlets after 14 days in the greenhouse.
 | Figure 2: Callus induction and plant regeneration in Vb-15. (a) 10-day-old cotyledon explant excised from in vitro germinated seed. (b) Friable callus with few differentiating shoots indicated by arrow. (c) Regenerating shoot indicated by arrow. (d) 8-week-old regenerants at the elongation stage. (e) Rooted plantlets. (f) 2-week-old potted out plantlet. [Click here to view] |
3.4. In vitro Response of Different Tomato Genotypes on MS + 0.5 mg-1TDZ
The callus induction and plant regeneration system developed [Figure 2] was evaluated on other tomato genotypes including Vb-15 as the reference genotype (control). Culturing cotyledons onto MS + 0.5 mg-1 TDZ from the ten genotypes tested resulted to 92–100% responding explants with signs of positive callus cell growth and rapid longitudinal expansion of the explant [Table 2]. Callus formation was observed only after 6 days of culture in Discovery, a commercially propagated tomato genotype. In most genotypes, dedifferentiation of callus was found at the embryo axis attachment to the hypocotyl, and at the wounded margins of the cotyledon. Although a high percentage of callus formation was observed (96–100%), there is a relatively slow callus growth in terms of CI where all genotypes formed callus only at one cut end or at both cut ends of the cotyledons. The medium optimized for Vb-15 has also stimulated shoot formation on other genotypes following different pathways. In tomato varieties Rosanna and Discovery, shoot has emerged directly without callus formation in 7–8 days. The two hybrid tomatoes have significantly different responses in terms of number of days and percent shoot formation. Shoot formation in Improved Apollo was observed in 21 days while in Apollo it was only 8 days. All varieties, except for Apollo, produced more shoots compared to Vb-15. For all Tm breeding lines tested, shoot formation was observed after 10–12 days. These Tm breeding lines were also superior to Vb-15 in terms of shoot formation. In general, all open-pollinated tomato genotypes have superior response in in vitro culture compared with hybrid tomatoes. From these results, the optimized medium for Vb-15 is reproducible and can be used for other genotypes. Moreover, the recalcitrance of Vb-15 in in vitro culture was also evident since the other genotypes produced more shoots with relatively higher responding explants per culture.
Table 2: Mean response of different tomato genotypes in terms of callus and shoot formation after 6 weeks of culture on MS+0.5 mg/L TDZ.
| Genotype | Responding explant (RE) (%)* | Callus formation/RE (%)* | Shoot formation/RE (%)* | No. of days to callus formation** | Mean callusing Index** | No. of days to shoot formation** |
|---|---|---|---|---|---|---|
| Vb-15 | 92 | 100 | 56.52 | 10.2±0.20c | 1.2±0.12b | 10.64±0.13cd |
| Improved Apollo | 92ns | 100ns | 78.26s | 10±0.0c | 1.32±0.13ab | 21±0.0f |
| Apollo | 100ns | 100ns | 44ns | 10±0.0c | 1.48±0.10ab | 11.18±0.35d |
| Discovery | 100ns | 100ns | 100s | 6.6±0.92a | 1.76±0.09a | 8.6±0.16b |
| Rosanna | 100ns | 96ns | 100s | 9±0.0b | 1.64±0.11ab | 7.8±0.08a |
| N. Red | 100ns | 100ns | 80s | 10±0.0c | 1.48±0.10ab | 7.75±0.10a |
| Tm 163147 | 100ns | 100ns | 100s | 10.4±0.40c | 1.64±0.10ab | 10.2±0.08c |
| Tm 12-18-0-8 | 100ns | 100ns | 80s | 10±0.0c | 1.48±0.10ab | 12±0.0e |
| Tm 14-1-1-5 | 92ns | 100ns | 65.22ns | 10±0.0c | 1.44±0.13ab | 12±0.0e |
| Tm 15-26-0-10 | 96ns | 100ns | 79.17s | 10±0.0c | 1.28±0.11ab | 12±0.0e |
Significant (s) or not significant (ns) in comparison with the reference genotype Vb 15 using binary logistic regression analysis.
Means±standard error with the same letter is not significantly different using least significance difference, RE: Responsive explants.