Peanut (Arachis hypogaea L.) production has faced a notable decline worldwide due to multiple environmental and agronomic challenges. Among these, drought conditions and nitrogen (N) deficiency are particularly significant constraints, especially in regions where water availability is limited, and soil is nutrient-poor. Adequate irrigation and balanced nitrogen supply are critical to maintaining optimal plant growth and yield potential [1,2]. In recent years, native endophytic microorganisms (NEMs) have gained attention for their ability to enhance crop resilience and productivity under such stress conditions.
NEMs are a diverse group of beneficial microbes including bacteria, actinomycetes, and fungi that reside within plant tissues for part or all their life cycles without causing harm to the host. These microorganisms contribute to plant health through both symbiotic and non-symbiotic mechanisms, such as nutrient solubilization, nitrogen fixation, hormone production, and disease suppression [3-7]. Their colonization of root systems supports improved nutrient uptake and mitigates the effects of biotic and abiotic stresses. Notably, the integration of NEM into cropping systems has demonstrated the potential to significantly reduce the dependency on CNFs [8,9]. For example, studies in Zea mays (maize) cultivation have shown that supplementing NEMs can cut nitrogen fertilizer usage by 50% without compromising yield. In some instances, the quality of maize cobs produced with NEM supplementation was comparable to or even superior to that of crops grown using full doses of synthetic nitrogen inputs [10,11]. In addition to yield stability, the use of NEM improved key soil fertility indicators such as increased pH, organic matter, total nitrogen (TN) content, and available phosphorus (AP) while simultaneously enhancing plant physiological traits such as height, leaf number, and chlorophyll concentration. In a related study, the application of Bacillus NTLG2-20 resulted in a 4.0% higher fresh cob yield in baby corn when compared with Bacillus aryabhattai strain CM44, and a 12.7% improvement relative to the uninoculated control. Furthermore, pre-sowing inoculation of seeds with B. aryabhattai strain CM44 led to a 9.02% yield increase over non-inoculated treatments, emphasizing the effectiveness of microbial inoculation strategies [10,12]. Similar benefits were observed when NEM application was combined with a 50% reduction in chemical nitrogen inputs. This integrated approach achieved fresh cob yields statistically equivalent to the conventional 100% nitrogen fertilizer treatment, demonstrating the feasibility of reducing chemical fertilizer reliance while sustaining crop productivity [13,14].
In peanut cultivation, NEMs based strategies have also been explored for their role in enhancing tolerance to salt stress and improving water use efficiency. When applied together with organic and mineral fertilizers, Bacillus species inoculants helped mitigate salt-induced damage and improved chlorophyll content, further contributing to plant vigor and yield [15]. Beyond abiotic stress mitigation, NEMs have shown promise in managing soil-borne diseases, a critical concern in peanut production systems. One of the most destructive pathogens in peanut fields is fungus Sclerotium rolfsii Sacc. responsible for causing southern stem and root rot. In Vietnam, it is estimated that over 25% of peanut crops are affected by this fungal disease [16,17]. S. rolfsii is notoriously difficult to manage due to its broad host range infecting more than 500 plant species and its production of persistent sclerotia that can survive in the soil for years without a host [18,19]. As a result, biological control methods have been increasingly explored as sustainable alternatives to chemical fungicides. Research has shown that Streptomyces and Bacillus genera exhibit strong antagonistic activity against S. rolfsii. These microbial strains not only reduce the incidence of stem and root rot but also enhance crop performance through their ability to fix nitrogen and promote plant growth [20-24]. In experimental trials, treatments involving either Streptomyces sp. or Bacillus sp. led to a significant reduction in disease occurrence compared to non-inoculated controls. Interestingly, while combined inoculation of both strains was effective in disease suppression, it did not significantly outperform individual inoculations in terms of total fruit count or chili yield [25].
Given these promising outcomes, the current research aims to evaluate the effectiveness of strain cjy13 and strain CAU 1564 in enhancing peanut productivity while reducing the need for synthetic nitrogen inputs. Conducted over two consecutive growing seasons, the study seeks to compare the effects of individual and combined inoculations of these strains on soil fertility, plant growth, and final yield. The goal is to identify optimal microbial treatment strategies that can sustainably improve legume production, reduce environmental impacts of fertilizer overuse, and contribute to long-term agricultural resilience. By integrating NEMs into nutrient management plans, farmers can achieve more efficient use of natural resources, improved soil health, and enhanced crop yields all while minimizing the ecological footprint of conventional agricultural practices. Continued research in this field is essential for developing location specific, crop specific microbial solutions that can address the multifaceted challenges of modern farming systems.
2. MATERIALS AND METHODS
2.1. Experimental Location and Time
The field site was conducted at the Agricultural Experiment Center of An Giang University from July to October in 2024 (crop 1), and November to February in 2025 (crop 2).
2.2. Experimental Design
The experiment was arranged in a randomized complete block design (RCBD) with nine treatment plots; each replicated 4 times. The treatments included: (1) strain cjy13 + 0.0 kg N/ha, (2) strain CAU 1564 + 0.0 kg N/ha, (3) strain cjy13 + strain CAU 1564 + 0.0 kg N/ha, (4) strain cjy13 + 20.0 kg N/ha, (5) strain CAU 1564 + 20.0 kg N/ha, (6) strain cjy13 + strain CAU 1564 + 20.0 kg N/ha, (7) strain cjy13 + 40.0 kg N/ha, (8) strain CAU 1564 + 40.0 kg N/ha, and (9) strain cjy13 + strain CAU 1564 + 40.0 kg N/ha. Urea, superphosphate, and potassium chloride were applied separately, with nutrient contents converted to their equivalent amounts of nitrogen (N), phosphorus (P), and potassium (K) in kg/ha, not exceeding the maximum recommended rates for peanut cultivation in the study area (40 kg N, 60 kg P, and 60 kg K/ha) as suggested by local farmers and Chuong [26]. The total experimental area was 720 m², with each plot measuring 1.0 m in width and 20.0 m in length. Plots were arranged with 250 cm spacing between replications and 25 cm between planting holes, with two seeds sown per hole in single rows. After the third leaf stage, only one healthy seedling per hole was maintained. Initial soil analyses revealed pH values of 6.7 (2024) and 6.5 (2025), regarding cation exchange capacity (CEC) of 4.23 and 5.21 cmol+/kg, soil organic matter (SOM) at 1.83% and 2.34%, TN at 0.125% and 0.267%, and available phosphorus at 254 and 280 mg/kg. Exchangeable potassium was undetectable in both years. The soil was classified as sandy loam, with textures comprising silt (18.0% and 19.0%), sand (80.0% and 77.9%), and clay (2.0% and 3.1%) based on USDA standards [27], and deemed suitable for peanut cultivation [28].
2.3. Microorganisms Source
Strain cjy13 and strain CAU 1564 were isolated from peanut root nodules. Strain cjy13 colonies were isolated from nodulated peanut roots 65 days after sowing (DAS) on yeast mannitol agar (YMA) [29], while strain CAU 1564 colonies were isolated using Streptomyces Project Agar Number 2 (ISP2) [30]. It served as the medium for isolating and culturing Streptomyces species. After autoclaving and cooling to 40°C, the medium received supplements of cycloheximide (50 μg/mL), nystatin (40 μg/mL), and nalidixic acid (54.9 μg/mL) to prevent contamination from other bacteria and fungi. Two bacterial suspensions, each containing strain cjy13 and strain CAU 1564 with concentrations reaching 108 CFU/mL, were characterized using molecular techniques and phylogenetic tree analysis [Figure 1] conducted at Nam Khoa Company in Ho Chi Minh City, Vietnam. For 16S ribosomal DNA (rDNA) sequencing, pure ENFB colonies were transferred to Eppendorf tubes. DNA was extracted from these samples utilizing Thermo Scientific™ GeneJET Genomic DNA Purification Kits. The 16S rDNA genes of the isolated strains were subsequently amplified through PCR with the primer set 20F (5′-CTACGGCAAGGCGACGCTGACG-3′) and 1500R (5′-GGTTACCTTGTTACGACTT-3′) [31,32]. Sequence data underwent analysis using MEGA software and were compared against the most similar sequences in GenBank through the BLAST technique, with an identity threshold exceeding 99.70% [33]. Biochemical assays of strain cjy13 (cjy13) confirmed strong nitrogen-fixing activity in liquid YMA medium and ISP2 for strain CAU 1564 after 6 days of incubation. While, for more information, please see in the attached supplementary figure file.
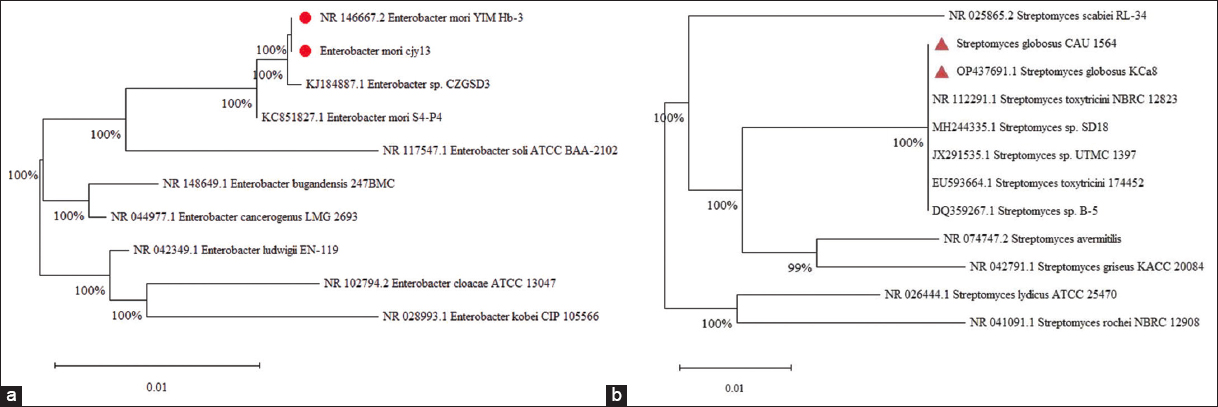 | Figure 1: Phylogenetic tree of partial 16S ribosomal RNA sequences of (a) strain cjy13 and (b) strain CAU 1564 were isolated from peanut nodules along with the sequences from selected references strains. The tree was constructed by the neighbor-joining method using MEGA 11. The scale bar corresponded to 0.01 substitutions per nucleotide position. Numbers at the branches were bootstrap percentages. [Click here to view] |
2.3.1. Thermal tolerance assessment
The ability of the strain cjy13 to withstand elevated temperatures was evaluated in two phases. In the first step, YMA nutrient broth was prepared in test tubes for each strain. In the second step, the strains were streaked onto YMA agar slants and incubated at 25, 37, and 45°C. Each treatment included four replicates, and colony growth was observed over a period of 7 days [34].
2.3.2. Salt and pH tolerance
To determine salt tolerance of strain cjy13, liquid YMA medium was supplemented with NaCl to final concentrations of 0.5%, 2%, 3%, 4%, and 5%. Colonies of the four strains were individually inoculated into these media and incubated at 28°C. Each treatment was performed with four replicates, and growth responses were recorded after 1 week [34].
To determine pH tolerance of strain cjy13, liquid YMA medium was adjusted to 4.5 with 0.1 N HCl, and to 7.0 and 8.5 with 0.1 N NaOH, followed by verification with a calibrated pH meter. Inoculated cultures were incubated at 28 °C with four replicates for each level, and bacterial growth was assessed after 7 days [34].
2.3.3. Ammonia production
To determine salt tolerance of strain cjy13, Ammonia production capacity was assessed qualitatively by inoculating each strain into peptone water and incubating on YMA at 30 °C for 60–80 h. The presence of ammonia was confirmed by a color shift from brown to yellow following the addition of Nessler’s reagent [35].
2.3.4. Nitrogenase activity
Nitrogenase activity was determined using the acetylene reduction assay as described by Soper et al. [36]. To determine nitrogenase activity of strain cjy13, initially grown in YMA broth for 24 h, and the resulting suspensions were transferred into nitrogen-free liquid medium. A control (N-free medium without inoculation) was included. Cell density was adjusted to OD600 = 0.8 under incubation at 30°C and 160 rpm [34,36].
2.3.5. Nitrogen concentration analysis
For nitrogen quantification, strain cjy13 was cultured in nitrogen-free medium containing 0.05% malate as the carbon source and incubated at 30°C. After centrifugation at 3000 rpm for 1 min, the supernatant was collected and analyzed according to Daoliang et al. [37].
2.4. Cultivation and Spore Preparation
Enterobacter mori strain cjy13 was cultured in YMA medium and incubated at 27°C for 24 h with continuous shaking at 150 rpm to obtain actively growing cell suspensions, following standard procedures for Gram-negative bacteria [34]. Streptomyces globosus strain CAU 1564 was cultivated on diluted ISP2 medium and incubated at 27 °C for 5 days to allow sporulation. Mature spores were harvested by flooding the agar surface with sterile distilled water, gently scraping, and filtering through sterile cotton to remove mycelial fragments (Shirling and Gottlieb, 1966; Seipke et al., 2012). The resulting cjy13 cell suspension and CAU 1564 spore suspension were then adjusted to the desired density for subsequent inoculation experiments [38].
2.5. Spore Collection and Seed Inoculation
Spores of both strains were harvested by centrifugation at 10,000 × g for 5 min at 4 °C. The resulting pellets were washed 3 times with sterile distilled water to remove residual medium components. After washing, the spore suspensions were resuspended in sterile distilled water and adjusted spectrophotometrically to approximately 108 CFU/mL. All seeds were thoroughly soaked before inoculation. After inoculation, the treated seeds were incubated in the dark for approximately 8 h before planting, with the final colony counts of both strains reaching 108 CFU/seed. For the control treatment, seeds were soaked with sterile distilled water instead of bacterial inoculum.
2.6. Field Planting and Additional Inoculation
Peanut seeds (L14) were used for the field experiment. They were planted in single rows with the following spacing: (i) 20 cm between individual plants; (ii) 30 cm between small rows; and (iii) 50 cm between large rows. To ensure sufficient microbial presence, two additional inoculations were performed on the peanut plants after 20 and 45 DAS.
2.7. Material, Fertilizers, and Irrigation Water
The peanut variety L14 was collected from Vietnam Agricultural Sciences Institute, Vietnam and used for this study. This variety is commonly grown in the research area. It has high yield potential, is resistant to pests and diseases, and has a growth duration of approximately 100 days.
The chemical fertilizers used were urea, superphosphate, and potassium chloride produced by Binh Dien Fertilizer Company, Vietnam. Chemical fertilizers were applied in three stages as follows: (i) Basal application: the full dose of phosphorus fertilizer combined with one-third of urea and one-third of KCl; (ii) First topdressing (at the three-true-leaf stage): one-third of urea and one-third of KCl; and (iii) Second topdressing (25–30 DAS): the remaining quantities of fertilizers.
Deep well water was used for irrigating peanut plants throughout the experiment. Under non-rainfall conditions, irrigation was carried out twice daily, in the morning and afternoon. During the flowering stage, supplemental watering was applied to maintain adequate soil moisture. The irrigation method employed was uniform spraying across the peanut field.
2.8. Data Collection and Analytical Methods
The study recorded plant growth parameters over 2 years, including plant height, total chlorophyll content, shoot number, and leaf count, all measured at 65 DAS from an average of 20 healthy plants per treatment during the crop 1 and crop 2 growing seasons. Yield traits assessed included nodule count and weight, total biomass, number of filled and unfilled pods, 1,000-seed weight, and fresh pod yield. Nutritional analyses, including moisture, lipid, protein, nitrogen, phosphorus, and potassium content, were performed following the methods outlined by Delahaut and Marega [39]. Soil physical and chemical characteristics were evaluated according to procedures described by Arnold [40] and the USDA [27].
2.9. Statistical Analysis
All data were processed using Statgraphics XVI software for analysis of variance. Comparisons among treatment means were made using Duncan’s multiple range tests at a significant level of P ≤ 0.05.
3. RESULTS
3.1. Assessment of Morphological Traits and Biochemical Properties of Strain Cjy13
Table 1 shows that the biochemical characterization of strain cjy13 revealed a diverse metabolic profile with strong adaptability to environmental conditions. The strain efficiently utilized several carbohydrates, including glucose, mannose, mannitol, raffinose, and esculin, while failing to metabolize galactose and ribose, indicating substrate selectivity. Positive reactions for starch hydrolysis, β-glucosidase, and glucose oxidation suggest the presence of extracellular enzymes that facilitate the breakdown of complex polysaccharides. Strong catalase activity reflects the ability to counter oxidative stress, whereas the absence of oxidase activity confirms its metabolic distinction from certain aerobic bacteria. The strain also displayed nitrate reduction, citrate utilization, and a positive Voges-Proskauer reaction, highlighting versatile nitrogen and carbon metabolic pathways. Furthermore, growth was maintained across a wide range of salinity (0.5–5% NaCl), pH (4.5–8.5), and temperature (25–45°C), demonstrating robust environmental tolerance. Collectively, these traits support the potential of E. mori cjy13 as a resilient and functionally diverse endophytic bacterium.
Table 1: The physical and chemical characterization of E. mori cjy13.
| Biochemical test | E. mori cjy13 | Biochemical test | E. mori cjy13 |
|---|---|---|---|
| Beta–Xyloidine | + | NaCl (0.5–5%) | ++ |
| D–Mannose | + | Temperature (25–45°C) | ++ |
| D–Glucose | + | pH (4.5–8.5) | ++ |
| D–Galactose | 2212 | Citrate use | + |
| D–Ribose | − | Β–Glucosidase | + |
| Catalase | ++ | Mannitol | + |
| Oxidase | − | Raffinose | + |
| Starch hydrolysis | + | Glucose oxidation | + |
| Esculin hydrolysis | + | Nitrate reduction | + |
| Methyl-Red | − | Voges-Proskauer | + |
| Ornithine Decarboxylase | + |
(−): Negative reaction; (+): Weak reaction; (++): Strong reaction. E. mori: Enterobacter mori
Figure 2 shows that the activity of nitrogenase was evaluated using the acetylene reduction assay, in which acetylene (C2H2) is converted to ethylene (C2H4). This was performed with a C2H2 gas injection system. After the initial addition of acetylene, a short lag phase occurred before the reaction stabilized, showing consistent performance throughout a 72-h incubation. The reduction rate was determined from the linear accumulation of ethylene observed during this steady phase. All experiments were carried out under controlled laboratory conditions with four replications per treatment.
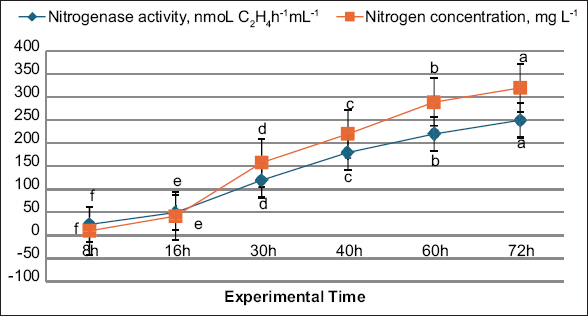 | Figure 2: Nitrogenase activity of strain L-1 as assessed by acetylene reduction and nitrogen content. Distinct letters (a-f) denote statistically significant differences at P < 0.01. [Click here to view] |
As illustrated in Figure 2, the nitrogenase activity of strain cjy13 was successfully quantified. The strain showed a progressive rise in activity across the incubation period, peaking at 320 nmol C2H4 h−1 mL−1 at 72 h. A parallel increase was also noted in TN content, measured through the Kjeldahl method. Nitrogen concentration rose from an initial 10 mg N/L to a maximum of 250 mg N/L. These variations were statistically significant at the 1% level. The results highlight the ability of E. mori cjy13 to substantially enhance nitrogen fixation, suggesting its strong potential as an endophytic inoculant for sustainable biofertilizer applications.
3.2. Interaction Effects of Microbial Strains and Nitrogen Fertilizer on Soil Chemical Properties
Table 2 demonstrated that both microbial inoculants and chemical nitrogen fertilizers significantly influenced key soil properties, including pH, CEC, SOM, TN, and AP over the two-crop period (2024–2025). For microbial treatments (Factor A), co-inoculation of strain A and strain B resulted in the highest soil pH in crop 2 (6.15), significantly greater than either strain applied individually (P < 0.01). This suggests synergistic effects on buffering soil acidity, possibly due to improved microbial interactions and organic acid regulation. Analysis of variance revealed significant interaction effects (P < 0.01 or P < 0.05) between microbial strains (factor A) and CNF levels (factor B) on most soil chemical parameters across both cropping seasons, except for SOM, where no significant interaction was observed.
Table 2: Effects of strain cjy13 and strain CAU 1564 combined with CNF rates on soil chemical properties on both crops.
| Factors | pH | CEC (Cmol+/kg) | SOM (%) | TN (%) | AP (mg/kg) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | |
| NEM (108CFU/mL) (A) | ||||||||||
| Strain cjy13 | 6.02b | 5.52c | 5.90a | 6.90a | 2.31b | 3.71b | 0.26a | 0.30a | 272c | 296b |
| Strain CAU 1564 | 6.41ab | 5.91b | 5.43ab | 5.83b | 2.54a | 3.94a | 0.21c | 0.22c | 296a | 312a |
| Strain (cjy13+CAU 1564) | 6.65a | 6.15a | 5.23b | 5.83b | 2.31b | 3.51c | 0.25ab | 0.26ab | 280b | 290b |
| CNF (kg N/ha) (B) | ||||||||||
| 0.00 | 5.76c | 5.26b | 3.29b | 3.19b | 2.32 | 2.12 | 0.18b | 0.08c | 229 | 219a |
| 20.0 | 6.67a | 6.17a | 5.24ab | 5.64ab | 2.31 | 2.11 | 0.20ab | 0.13b | 229 | 209b |
| 40.0 | 6.10b | 6.05ab | 5.54a | 5.94a | 2.30 | 2.12 | 0.22a | 0.19a | 220 | 190c |
| F (A) | ** | ** | ** | ** | ** | ** | ** | ** | ** | ** |
| F (B) | ** | ** | ** | ** | ns | ns | * | * | ns | * |
| F (AxB) | ** | ** | ** | ** | ns | ns | ns | ** | ns | * |
| CV (%) | 11.8 | 11.5 | 24.3 | 12.3 | 11.6 | 11.6 | 18.7 | 15.7 | 12.7 | 11.7 |
Soil samples were collected at harvest per crop. There were four samples per treatment for nine treatments, totaling 36 soil samples per season. The total number of soil samples for both seasons was 72.
The interaction between the two factors markedly influenced soil pH. The combination of strain cjy13 with strain CAU 1564 consistently maintained higher pH values, especially at lower nitrogen levels (0 and 20 kg N/ha), suggesting a potential buffering effect against soil acidification. Regarding CEC, the interaction effect was evident, with treatments involving strain cjy13 or strain CAU 1564 in combination with 40 kg N/ha resulting in the highest CEC values in Crop 1. However, these effects were less pronounced in Crop 2, indicating temporal variability in microbial activity and soil response. For TN and AP, interaction effects were also significant. At the highest nitrogen level (40 kg N/ha), strain A alone tended to maintain higher TN and AP values, while the combined the strain cjy13 and strain CAU 1564 inoculation did not show synergistic benefits and, in some cases, resulted in lower nutrient availability. This may indicate potential antagonistic interactions or competition between the strains under certain nutrient conditions.
Although no significant interaction was found for SOM, strain CAU 1564 consistently resulted in higher SOM content than the other treatments across both seasons, highlighting its potential role in enhancing SOM accumulation. These findings underscore the complexity of microbial fertilizer interactions and emphasize the importance of considering both biological and chemical inputs when aiming to optimize soil fertility and nutrient availability.
Moreover, this combination maintained a relatively stable CEC over time, indicating enhanced nutrient retention capacity in the soil. Regarding SOM, the treatment with strain cjy13 alone yielded the highest content in crop 2 (3.94%), outperforming the co-inoculation. Interestingly, TN was more influenced by strain cjy13, with the highest value observed in its sole application. However, AP was significantly improved by strain CAU 1564, possibly due to its phosphate-solubilizing abilities.
For chemical nitrogen fertilizer (Factor B), increasing nitrogen rates generally decreased AP, especially at 40.0 kg N/ha, where AP dropped to 190 mg/kg in crop 2. Conversely, TN increased with higher fertilizer rates, with the maximum at 40.0 kg N/ha. Interaction effects of microbial inoculation with CNF ratios were significant for most parameters, including TN and AP (P < 0.05), suggesting that microbial activity can modify the outcome of chemical inputs, thereby supporting integrated nutrient management strategies.
3.3. The Influences of Strains cjy13 and CAU 1564 Combined with CNF Rates on Peanut Growth
Figure 3 presents the impact of microbial inoculation and nitrogen fertilization on peanut plant height and branch number at 65 DAS during both experimental crops. The analysis revealed statistically significant variations, with plant height and branch number differing by 5.0% and 1.0%, respectively, across treatments in both years. Notably, the application of strain cjy13 or strain CAU 1564 individually led to significant improvements in both growth parameters when compared to the co-inoculated treatment. The best performance was observed in Treatment 4 (strain A combined with 20 kg N/ha), indicating a positive synergistic effect between moderate nitrogen input and single-strain inoculation. Conversely, plants treated with the combined inoculation of strain cjy13 and strain CAU 1564 showed the lowest values for both height and branch number.
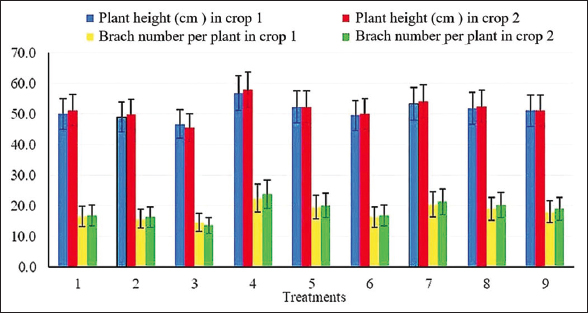 | Figure 3: Effects of Enterobacter mori cjy13 and Streptomyces globosus CAU 1564 on average plant height and branch number at 65 days after sowing (DAS) in both experimental years. Note: Plant height and branch number were monitored on 20 plants per replicate at 65 DAS for both 2024 and 2025. [Click here to view] |
Figure 4 shows the effects of microbial inoculation and nitrogen fertilizer levels on leaf number and total chlorophyll content in peanut plants at 65 DAS. Significant differences were detected at both the 5% and 1% levels, confirming strong treatment effects. The highest values occurred in Treatment 4, where strain cjy13 was applied with 20 kg N/ha, reaching 115 and 124 leaves per plant in the first and second years, respectively, while chlorophyll content peaked at 44.9 and 46.3 mg m−2. Overall, both leaf number and chlorophyll content were slightly higher in the second crop compared with the first, suggesting a cumulative benefit of microbial inoculation. In contrast, the lowest values were observed in Treatment 3, with co-inoculation of strains cjy13 and CAU 1564 under zero-N conditions. Taken together, the results indicate that individual inoculation with either strain cjy13 or strain CAU 1564, when combined with moderate CNF, was more effective in promoting vegetative growth traits than their combined application
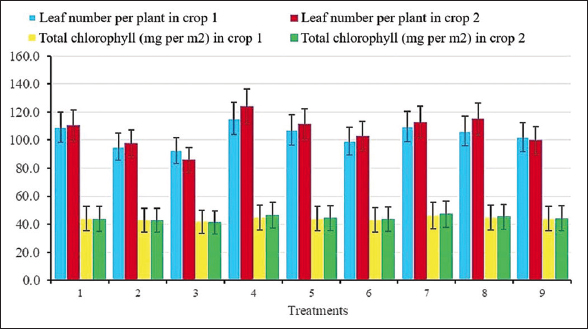 | Figure 4: Effects of Enterobacter mori cjy13 and Streptomyces globosus CAU 1564 on average leaf number and total chlorophyll at 65 days after sowing (DAS) in both experimental years. Total chlorophyll and leaf number were monitored on 20 plants per replicate at 65 DAS for both 2024 and 2025. [Click here to view] |
3.4. Effects of Strain cjy13 and CAU 1564 Combined with CNF Rates on Peanut Yield Components and Yield
Table 3 highlighted the inoculated effects of strain cjy13 and strain CAU 1564, along with varying CNF rates, on peanut yield components. The results indicate that inoculation with either strain cjy13 or strain CAU 1564 alone resulted in a higher number of filled pods per plant compared to the co-inoculation of both strains. In contrast, the combined application of both microbial strains led to a reduction in filled pod number, suggesting potential antagonistic interactions that may have interfered with nutrient uptake or pod development. Interestingly, treatments with 50% CNF applied alongside either strain cjy13 or strain CAU 1564 showed no significant difference in filled pod number when compared to full CNF application, suggesting that NEM inoculation can maintain yield levels even with reduced nitrogen inputs. In addition, unfilled pod numbers were lower in single-strain inoculations than in combined treatments, further supporting the superior effectiveness of individual microbial applications. The data presented in Table 4 demonstrate the influence of microbial inoculation and nitrogen fertilizer application on peanut yield components over two consecutive crops. Notably, strain cjy13 inoculation combined with 20 kg N/ha (Treatment 4) consistently produced the highest number of filled pods and pod weights in both crops. In crop 2, this treatment achieved 90.3 pods per plant and 177 g per plant in pod weight, suggesting a strong synergistic effect between moderate nitrogen input and microbial enhancement. In contrast, treatment 3, which involved co-inoculation of strain cjy13 and strain CAU 1564 without CNF, resulted in significantly lower values for filled pod number and weight, and higher numbers of unfilled pods.
Table 3: Effects of strain cjy13 and strain CAU 1564 combined with CNF rates on yield traits of peanuts in both crops.
| Treatments | No. of filled pods (pod/plant) | Filled pod weight (g/plant) | No. of unfilled pods (pod/plant) | |||
|---|---|---|---|---|---|---|
| Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | |
| 1 | 74.5a | 66.5ab | 160ab | 169ab | 55.8abc | 6.75bc |
| 2 | 68.4a | 57.8bc | 163ab | 161ab | 57.8abc | 9.25ab |
| 3 | 41.9b | 34.8c | 127b | 94.7b | 84.0a | 11.0a |
| 4 | 72.1a | 90.3a | 207a | 177a | 35.0c | 4.75c |
| 5 | 66.8a | 75.0ab | 189a | 175a | 56.5abc | 6.25bc |
| 6 | 59.5ab | 65.3ab | 156ab | 150b | 51.5bc | 6.5bc |
| 7 | 74.5a | 62.8b | 191a | 179a | 53.0bc | 4.75c |
| 8 | 76.6a | 52.5bc | 187a | 183a | 47.0bc | 7.5abc |
| 9 | 57.7ab | 69.3ab | 111b | 165ab | 69.8ab | 8.5ab |
| F-test | * | * | * | * | * | * |
| CV (%) | 22.2 | 23.9 | 31.1 | 26.0 | 29.8 | 31.2 |
Pod samples were collected at harvest per crop. There were four samples per treatment across nine treatments, totaling 36 samples per crop. The total number of samples for each indicator for both crops was 72. Values within the same column followed by different letters are significantly different at P<0.05 (*).CNF: Chemical nitrogen fertilizer, CV: Coefficient of variation
Table 4: Effects of strain cjy13 and strain CAU 1564 combined with CNF rates on peanut yield traits in both crops.
| Treatment | Wt. of 100 pods (g) | Wt. of 1,000 seeds (g) | Fresh pod yield (t/ha) | |||
|---|---|---|---|---|---|---|
| Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | |
| 1 | 93.3bc | 107bc | 316b | 313cd | 5.00ab | 5.11b |
| 2 | 90.0c | 103bc | 266d | 300de | 4.92ab | 5.15b |
| 3 | 82.0d | 76.7d | 266d | 250f | 3.19b | 3.82c |
| 4 | 113a | 120a | 333a | 383a | 5.88a | 6.54a |
| 5 | 100b | 110abc | 300c | 343bc | 5.06ab | 5.16b |
| 6 | 90.0c | 105bc | 266d | 323bcd | 4.33bc | 4.82bc |
| 7 | 110a | 114ab | 333a | 360ab | 5.83a | 6.35a |
| 8 | 96.5bc | 109abc | 300c | 333bcd | 5.05ab | 5.20b |
| 9 | 91.6c | 100c | 266d | 273ef | 4.53bc | 4.81bc |
| F-test | ** | ** | ** | ** | * | ** |
| CV (%) | 11.1 | 13.2 | 9.54 | 14.4 | 23.3 | 18.2 |
Pod and seed samples were collected at harvest per crop. There were four samples per treatment across nine treatments, totaling 36 samples per crop. The total number of samples for each indicator for both crops was 72. Values within the same column followed by different letters are significantly different at P≤0.05 (*) and P≤0.01 (**).CNF: Chemical nitrogen fertilizer
3.5. Effects of Strains cjy13 and CAU 1564 Combined with CNF on Peanut Nutrition Composition
Table 5 presented the effects of two strains, which concluded strain cjy13 and strain CAU 1564 on the average nutrient composition of peanut seeds during the two experimental crops in 2024 and 2025. The results indicated statistically significant differences (P ≤ 0.01) among treatments in nutrient compositions of peanut seeds such as lipid, protein, nitrogen, phosphorus, and potassium. Treatment 4, which likely involved a combination of both these strains, produced the most notable results, with the highest lipid content (28.8% in crop 1 and 29.5% in crop 2), along with elevated phosphorus (0.312–0.33%) and potassium levels (0.327–0.515%), suggesting its potential in improving seed nutritional quality. Interestingly, treatment 7 recorded the highest protein content (19.5–19.6%) and nitrogen levels (3.12–3.14%), indicating the beneficial effects of strain CAU 1564 or its synergistic use. Conversely, treatments 3 and 9 showed the lowest performance across most parameters, particularly in protein and lipid content, which implies they may represent control groups or treatments without microbial inoculation.
Table 5: Effects of strain cjy13 and strain CAU 1564 combined with CNF rates on nutrient composition of peanut seeds in both crops.
| Treatments | Average values of peanut seed composition in crop 1 and crop 2 (%) | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Lipid | Protein | Nitrogen | Phosphorus | Potassium | ||||||
| Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | Crop 1 | Crop 2 | |
| 1 | 26.8bc | 27.2b | 18.6ab | 18.9ab | 2.98b | 3.02bc | 0.280c | 0.285bcd | 0.415ab | 0.475abcd |
| 2 | 25.7cd | 25.6c | 17.6bcd | 18.0bc | 2.82d | 2.88cd | 0.291bc | 0.312bc | 0.421ab | 0.442efg |
| 3 | 23.7e | 24.5de | 15.9e | 16.0e | 2.54g | 2.56e | 0.289bc | 0.27cd | 0.433a | 0.455cdef |
| 4 | 28.8a | 29.5a | 17.9bcd | 19.5a | 2.87c | 3.04a | 0.312a | 0.33ab | 0.327c | 0.515a |
| 5 | 26.7bc | 27.1b | 17.3cd | 18.4bc | 2.77e | 2.94bcd | 0.314a | 0.329ab | 0.438a | 0.492abc |
| 6 | 24.9de | 25.5cd | 17.0d | 17.8cd | 2.72f | 2.85cd | 0.296ab | 0.296bcd | 0.427a | 0.395g |
| 7 | 27.4ab | 27.9b | 19.5a | 19.6a | 3.12a | 3.14ab | 0.302ab | 0.378a | 0.418ab | 0.502ab |
| 8 | 25.4cd | 25.8c | 18.3bc | 18.5bc | 2.93c | 2.96bcd | 0.294bc | 0.290bcd | 0.428a | 0.445def |
| 9 | 23.7e | 23.8e | 16.9de | 17.0d | 2.70f | 2.72de | 0.260d | 0.260d | 0.395b | 0.421fg |
| F-test | ** | ** | ** | ** | ** | ** | ** | ** | ** | ** |
| CV (%) | 7.34 | 6.88 | 6.83 | 6.88 | 5.86 | 8.7 | 6.41 | 15.1 | 8.60 | 10.43 |
Seed samples were collected at harvest per crop. There were 4 samples per treatment across 9 treatments, totaling 36 samples per crop. The total number of samples for each nutrient indicator for both crops was 72. Values within the same column followed by different letters are significantly different at P<0.05 (*). Values within the same column followed by different letters are significantly different at P≤0.01 (**). CNF: Chemical nitrogen fertilizer, CV: Coefficient of variation
4. DISCUSSION
4.1. Integrating Microbial Inoculants and CNF Fertilization Effects on Soil Properties
This study demonstrated significant interactions between microbial inoculants (strain cjy13 and strain CAU 1564) and varying levels of CNF on key soil chemical properties, including pH, CEC, TN, and AP [Table 2]. These findings align with and are further elucidated by recent studies in the field. The observed maintenance of higher soil pH levels with combined inoculation of strain cjy13 and strain CAU 1564, especially at lower nitrogen rates, is consistent with findings by Zhou et al. [41], who reported that microbial inoculants can mitigate soil acidification caused by chemical fertilizers, thereby sustaining a more neutral pH conducive to nutrient availability and microbial activity. The enhancement of CEC observed in our study, particularly with strain cjy13 or strain CAU 1564 combined with 40 kg N/ha, mirrors result from Chuong [26], who found that microbial inoculants can increase CEC by promoting the accumulation of organic matter and enhancing soil structure, which, in turn, improves nutrient retention and exchange capacity. TN and available phosphorus: Our findings that strain A alone maintained higher TN and AP levels at the highest nitrogen rate, while the combination of strain cjy13 and strain CAU 1564 did not yield synergistic benefits, specific microbial strains can enhance nitrogen and phosphorus availability through mechanisms such as nitrogen fixation and phosphorus solubilization. However, they also noted that combining multiple strains does not always result in additive effects and may sometimes lead to antagonistic interactions, reducing overall efficacy [42]. SOM: Although no significant interaction was found for SOM in your study, the consistent increase in SOM with strain CAU 1564 inoculation aligns with Dang et al. [42], who reported that certain microbial inoculants contribute to organic matter accumulation by enhancing microbial biomass and activity, leading to increased residue decomposition and humus formation. These corroborative studies underscore the importance of selecting appropriate microbial inoculants and optimizing nitrogen fertilizer application rates to enhance soil chemical properties effectively. They also highlight the complex interactions between microbial communities and soil amendments, suggesting that tailored approaches are necessary for sustainable soil fertility management.
4.2. Effects of Two Microbial Strains and CNF Rates on Peanut Growth Parameters
On colonizing plant roots, NEM trains NEM establish a beneficial symbiotic relationship, adapting effectively to the nutrient-rich rhizosphere and contributing to host plant health [43-46]. These microbes support plant development by facilitating natural nitrogen fixation, supplying organic nitrogen to subsequent crops without synthetic input [47]. In addition, NEM enhances nutrient availability by transforming insoluble phosphorus into forms readily accessible to plants and by producing phytohormones like indole-3-acetic acid, which stimulate root elongation and nutrient uptake [29,48]. Similarly, members of the Streptomyces genus contribute significantly to soil health by decomposing organic matter and releasing nutrients such as phosphorus and potassium. Their ability to suppress soil-borne pathogens like root rot also supports plant resilience and productivity [49]. These microbial functions improve soil fertility and nutrient cycling, which are essential for achieving sustainable crop yields and maintaining long-term soil quality. Collectively, the integration of beneficial microbes into agricultural systems presents a promising strategy for reducing chemical inputs while enhancing both crop performance and environmental sustainability. This suggests potential microbial incompatibility or competitive interactions between the strains that may hinder plant development, as also highlighted by recent research emphasizing the importance of microbial compatibility in co-inoculation strategies [50,51]. Interestingly, strain B alone contributed to moderate growth enhancement, likely through its known roles in organic matter decomposition and phosphorus solubilization [49,51]. We observed that single-strain inoculations frequently outperformed combinations in this study. This outcome aligns with characteristics previously reported for this genus, where multi-strain approaches typically require proven synergistic interactions to be more effective [52]. Moreover, moderate nitrogen supplementation appears to complement microbial activity by supporting nutrient availability and plant vigor [53]. Therefore, targeted microbial applications combined with optimized nitrogen inputs can be an effective approach to enhancing peanut growth performance under field conditions. The decline in growth parameters under co-inoculation may stem from microbial competition or antagonism, reducing their overall effectiveness [47,54]. In addition, plant growth traits, including leaf number and chlorophyll content, were consistently higher in crop 1 than crop 2, likely due to improved soil conditions or accumulated microbial activity over time. In agreement with previous findings, beneficial endophytes like Streptomyces are known for producing a variety of growth-promoting substances and for suppressing plant pathogens, thereby enhancing plant health and vigor [12,34,55]. Seed inoculation with NEMs has been reported to steadily improve growth from sowing to harvest, supporting their use as a sustainable agricultural strategy [56].
4.3. Impact of Strains cjy13 and CAU 1564 Combined with CNF Rates on Peanut Yield Components and Yield
Regarding fruit weight, a consistent trend was observed: Both strain cjy13 and strain CAU 1564, when applied individually, resulted in significantly higher fruit weights compared to their combined inoculation. These findings underscore the importance of selecting compatible microbial strains for biofertilization strategies. Since this is the first time, these two strains have been tested, and their combined application in a field experiment is also novel, this discovery represents a key new finding of this research. Yield components and overall productivity were notably higher in crop 2 than crop 1, likely due to improved soil conditions or enhanced microbial establishment over time. As supported by Pratap et al. [57] and Kumar et al. [58], inoculating legumes with beneficial endophytes in nutrient-deficient soils enhances soil fertility, promotes plant development, and sustains yield performance, demonstrating the potential of microbial inoculants in low-input agriculture. This implies a potential antagonistic interaction between the two microbial strains when applied together without nitrogen support, possibly due to competition for root colonization or resources, which may have impaired nutrient uptake and pod development. Treatments involving both strain cjy13 or/and strain CAU 1564 generally outperformed the combined inoculation in terms of filled pod production, aligning with prior research indicating that microbial compatibility is essential for optimizing biofertilizer efficacy [55,59]. Furthermore, yield traits were slightly improved in crop 2, likely reflecting enhanced microbial establishment and improved soil conditions following initial inoculation [60]. These findings support the use of native endophytes, particularly in combination with reduced CNF application, as a sustainable strategy to maintain or even enhance peanut productivity under low-input conditions. These results support further exploration into the mechanisms underlying the improvement observed in plant productivity. The NEM inoculation enhanced the available nutrient uptake of peanuts and easier nutrient uptakes [61]. Endogenous bacteria could increase bacterial density in subsequent crops and might fix large amounts of natural nitrogen in the next season higher than the previous season. This is the reason why the amount of inorganic fertilizer is reduced annually [62].
4.4 Impact of Strains cjy13 and CAU 1564 Combined with CNF Rates on Peanut Nutrition
This study demonstrated significant interactions between microbial inoculants (strain cjy13 and strain CAU 1564) and varying CNF levels on key peanut yield components and seed quality parameters. These findings align with and are further elucidated by recent studies in the field. The observed increase in peanut yield with the combined application of microbial inoculants and reduced nitrogen fertilizer rates is consistent with findings from recent studies [43,63], who reported that inoculation with Bacillus sp. NTLG2-20, in conjunction with a 50% reduction in nitrogen fertilizer (20 kg N/ha), led to a 17.6% increase in peanut yield compared to no nitrogen application, without compromising yield relative to the full recommended nitrogen rate. This suggests that microbial inoculants can enhance nitrogen use efficiency, allowing for reduced fertilizer inputs without sacrificing yield. Seed quality improvement: our study’s findings of improved seed protein and lipid content with microbial inoculation are supported by research conducted by Ballat et al. [64], which demonstrated that microbial fertilizers produced by strains like Pantoea alhagi significantly increased peanut biomass and yield. The study highlighted that microbial additives could enhance nutrient uptake and assimilation, leading to improved seed composition. The notable increases in phosphorus and potassium in microbial treatments support findings from recent studies demonstrating the role of NEMs in nutrient mobilization and uptake [65]. Overall, these findings reinforce the hypothesis that beneficial microbes such as both strain cjy13 and strain CAU 1564 can enhance the nutrient content of leguminous crops, in alignment with recent research highlighting the positive impact of NEMs on crop productivity and soil fertility [53,66]. The inoculation crops with NEMs, such as strain cjy13 and strain CAU 1564, have been shown to significantly enhance plant growth and yield compared to non-inoculated controls. These endophytes promote plant development by facilitating nutrient uptake and improving physiological traits.
5. CONCLUSION
This study evaluated the effects of NEMs, particularly E. mori cjy13 and S. globosus CAU 1564, on peanut productivity, soil fertility, and seed quality across two growing seasons. Results suggest that inoculation of both strains can enhance soil health, yield, and nutritional quality compared with no CNF, with seed treatment using strain cjy13 indicating potential to maintain yield and quality under the locally recommended fertilizer regime of 20 kg N/ha, relative to the full CNF control. Individual inoculation with either strain outperformed co-inoculation, implying possible antagonism. Notably, improvements in soil fertility, yield components, and seed traits were more evident in the second crop, highlighting cumulative benefits of microbial use. While these findings demonstrate the potential of NEM-based strategies, particularly with strain cjy13 as a sustainable alternative to reduce CNF dependence, further validation through multi-site and long-term trials, supported by formal strain deposition, is essential to confirm broader applicability.
6. ACKNOWLEDGMENT
The authors express their gratitude to An Giang University for providing equipment support for this research through the Central Laboratory.
7. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
8. FUNDING
There is no funding to report.
9. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
10. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
11. DATA AVAILABILITY
All the data are available from the authors and shall be provided on request.
12. PUBLISHER’S NOTE
The views and statements presented in this article are entirely those of the authors and do not necessarily reflect the positions of the publisher, editors, or reviewers. The journal maintains a neutral stance on any jurisdictional claims related to institutional affiliations.
13. USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY
The authors confirm that no artificial intelligence (AI) tools were used in the writing or editing of this manuscript and that none of the images were altered using AI technology.
REFERENCES
1. Brasileiro AC, Morgante CV, Araujo AC, Leal-Bertioli SC, Silva AK, Martins AC. Transcriptome profiling of wild Arachis from water-limited environments uncovers drought tolerance candidate genes. Plant Mol Biol Rep. 2015;33:1876-92. [CrossRef]
2. Prasanna KB, Trimurtulu N, Gopal VA, Nagaraju Y. Impact of culturable endophytic bacteria on soil aggregate formation and peanut (Arachis hypogaea l.) growth and yield under drought conditions. Curr Microbiol. 2022;79(10):308. [CrossRef]
3. Trivedi P, Leach JE, Tringe SG, Sa T, Singh BK. Plant-microbiome interactions:From community assembly to plant health. Nat Rev Microbiol. 2020;18(11):607-21. [CrossRef]
4. Mukherjee A, Singh B, Verma JP. Harnessing chickpea (Cicerarietinum L.) seed endophytes for enhancing plant growth attributes and bio-controlling against Fusarium sp. Microbiol Res. 2020;237:126469. [CrossRef]
5. Mukherjee S, Siddiqui J, Saxena J. Microbial nanomaterials:Role in sustainable agricultural practices. Indian J Agric Sci 2024;94(5):451-60. [CrossRef]
6. Ali AR, Bahrami Y, Kakaei E, Elham K, Sara M, Sasan B. Isolation and identification of endophytic actinobacteria from Citrullus colocynthis (L.) Schrad and their antibacterial properties. Microb Cell Fact. 2022;21:1-17. [CrossRef]
7. Le CN, Hoang TK, Thai TH, Tran TL, Phan TP, Raaijmakers JM. Isolation, characterization and comparative analysis of plant-associated bacteria for suppression of soil-borne diseases of field-grown groundnut in Vietnam. Biol Control. 2018;121:256-62. [CrossRef]
8. Trang N, Chuong N. The enhancement of soil fertility and baby maize output by Streptomyces panayensis and vermicompost. Eurasian J Soil Sci. 2025;14(2):140-8. [CrossRef]
9. Padbhushan P, Sinha AK, Kumar U, Bhattachary PM, Poddar P. Plant growth-promoting bacteria and crop residue in rice-wheat system cultivated with favorable tillage influence crop productivity, nutrient uptake, soil quality, and profitability in the Terai Agro-ecological zone of West Bengal, India. Agronomy. 2023;13(10):2454. [CrossRef]
10. Chuong NV, Trang NN, Thuan NV. Nitrogen fertilizer use reduction by two endophytic diazotrophic bacteria for soil nutrients and corn yield. Commun Sci Technol. 2025;9(2):348-355. [CrossRef]
11. Ehinmitan AA, Losenge T, Mamati E, Ngumi V, Juma P, Siamalube B. BioSolutions for green agriculture:Unveiling the diverse roles of plant growth?promoting Rhizobacteria. Int J Microbiol. 2024;2024:618-91. [CrossRef]
12. Chuong NV. Influences of Enterobacter cloacae strain Fg 5-2 and its vermicompost and nitrogen fertilizer usage efficiency on groundnut yield. Trends Sci. 2024;21(9):8039. [CrossRef]
13. Van Chuong N. Influences of Enterobacter cloacae strain Fg 5-2 and its vermicompost and nitrogen fertilizer usage efficiency on groundnut yield. Eurasian J Soil Sci. 2024;23:133. [CrossRef]
14. Li Y, Chen Q, Zhang R. Effects of actinomycetes on soil fertility and plant growth:A review. Soil Biol Biochem. 2022;165:108540. [CrossRef]
15. Girna SL, Thales VA, Geocleber GS, Samuel OS, Francisco HR, Alisson GS, et al. Phosphate fertilization, biofertilizer and Bacillus sp. in peanut cultivation under salt stress. Rev Bras Eng Agric Ambient. 2024;28(4):e279003. [CrossRef]
16. Yan LY, Wang ZH, Song WD, Fan M, Kang YP, Lei Y, et al. Genome sequencing and comparative genomic analysis of highly and weakly aggressive strains of Sclerotium rolfsii, the causal agent of peanut stem rot. BMC Genomics. 2021;22:276. [CrossRef]
17. Barbosa AS, Sousa GG, Freire MH, Leite KN, Silva FD, Viana TV. Gas exchange and growth of peanut crop subjected to saline and water stress. Rev Bras Eng Agric Ambient. 2022;26:557-63. [CrossRef]
18. Wang H, Zhou L, Ma M. Interaction between nitrogen and phosphorus under different fertilization regimes:Implications for soil health. Agric Syst. 2023;206:103613. [CrossRef]
19. Jia S, Song C, Dong H, Yang X, Li X, Ji M, et al. Evaluation of efficacy and mechanism of Bacillus velezensis CB13 for controlling peanut stem rot caused by Sclerotium rolfsii. Front Microbiol 2023;16(14):1111965. [CrossRef]
20. Jacob S, Sajjalaguddam RR, Sudini HK. Streptomyces sp. RP1A-12 mediated control of peanut stem rot caused by Sclerotium rolfsii. J Integr Agric. 2018;17(4):892. [CrossRef]
21. Sousa JAJ, Olivares FL. Plant growth promotion by Streptomycetes:Ecophysiology, mechanisms and applications. Chem Biol Technol Agric. 2016;3(1):24. [CrossRef]
22. Yang N, Li LY, Sun BB, Xiao Y, Xu YP. Study on the effect of Bacillus amyloliquefaciens LX-J1 and its fertilizer on the control of penut white silk disease. Front Microbiol. 2017;25:73-6. [CrossRef]
23. Korangi AV, Argüelles AA, Ribeiro B, De Coninck B, Helmus C, Ongena M. Bacillus lipopeptide-mediated biocontrol of peanut stem rot caused by Athelia rolfsii. Front Plant Sci. 2023;14:1069971. [CrossRef]
24. Djordje F, Ivica D, Tanja B, Jelena L, Slaviša S. Biological control of plant pathogens by bacillus species. J Biotechnol. 2018;285:44-55. [CrossRef]
25. Azizah RU, Sri S, Triwidodo A, Sedyo H. Potential effect of rhizobacteria Streptomyces sp. and Bacillus sp. to geminivirus infection and red chili (Capsicum annum L.) quality production. J Agroteknol. 2023;14(1):43-52. [CrossRef]
26. Chuong NV. Effect of three different nitrogen rates and three rhizosphere N2- fixing bacteria on growth, yield and quality of peanuts. Trends Sci. 2024;21(3):7281. [CrossRef]
27. USDA. A Basic System of Soil Classification for Making sand Interpreting Soil Surveys (Handbook 436). Washington, DC:Natural Resources Conservation Service, U.S. Department of Agriculture;2019.
28. Rajendran G, Patel MH, Joshi SJ. Isolation and characterization of nodule-associated Exiguobacterium sp. from the root nodules of fenugreek (Trigonella Foenum-Graecum) and their possible role in plant growth promotion. Int J Microbiol. 2012;2012:693982. [CrossRef]
29. Chuong NV, Tri TL. Enhancing soil fertilizer and peanut output by utilizing endophytic bacteria and vermicompost on arsenic-contaminated soil. Int J Agric Biosci. 2024;13(4):596-602. [CrossRef]
30. Tian-Yi Y, Xiao-Liang L, Ya L, Xue-Wu S, Yong-Mei Z, Zheng-Feng Z, et al. Effect of phosphorus (P) on nitrogen (N) uptake and utilization in peanut. Acta Agron Sin. 2019;45:912-21.
31. Chibeba A, Kyei-Boahen S, Chibeba AM, Kyei-Boahen S, Guimarães MF, Nogueira MA, et al. Isolation, characterization and selection of indigenous Bradyrhizobium strains with outstanding symbiotic performance to increase soybean yields in Mozambique. Agric Ecosyst Environ. 2017;246:91-305. [CrossRef]
32. Del-Canto A, Sanz-Saez A, Sillero-Martínez A, Mintegi E, Lacuesta M. Selected indigenous drought tolerant rhizobium strains as promising biostimulants for common bean in Northern Spain. Front Plant Sci. 2023;14:1046397. [CrossRef]
33. Yarza P, Yilmaz P, Pruesse E, Glöckner FO, Ludwig W, Schleifer KH, et al. Uniting the classification of cultured and uncultured bacteria and archaea using 16S rRNA gene sequences. Nat Rev Microbiol. 2014;12(9):635-45. [CrossRef]
34. Van Chuong N, Le Kim Tri T. Isolation and Characterization identification of endophytic nitrogen-fixing bacteria from peanut nodules. Int J Microbiol. 2024;31:8973718. [CrossRef]
35. Borah A, Das R, Mazumdar R, Thakur D. Culturable endophytic bacteria of Camellia species endowed with plant growth promoting characteristics. J Appl Microbiol. 2019;127:825-44. [CrossRef]
36. Soper FM, Simon C, Jauss V. Measuring nitrogen fixation by the acetylene reduction assay (ARA):Is 3 the magic ratio. Biogeochemistry. 2021;152:345-51. [CrossRef]
37. Daoliang L, Xianbao X, Zhen L, Tan W, Cong W. Detection methods of ammonia nitrogen in water:A review. Trends Anal Chem. 2020;127:115890. [CrossRef]
38. Liu C, Wang X, Yan Y, Wang J, Zhang B, Zhang J, et al. Streptomyces heilongjiangensis sp. nov., a novel actinomycete that produces borrelidin isolated from the root surface of soybean [Glycine max (L.) Merr]. Int J Syst Evolut Microbiol. 2013;63(3):1030-6. [CrossRef]
39. Delahaut P, Marega R. Novel analytical methods in food analysis. Foods. 2022;11(10):1512. [CrossRef]
40. Arnold K. Methods of soil analysis:Part 1 physical and mineralogical methods. 2nd ed. Madison:The American Society of Agronomy, Inc.;1986. 1172.
41. Zhou X, Zhang J, Huang D. Synergistic effects of microbial inoculants on soil quality and crop productivity:A meta-analysis. Appl Soil Ecol. 2021;165:103982.
42. Dang PTH, Chuong NV. Priestia megaterium Thr45 reduces nitrogen and potassium fertilizer inputs while enhancing soil fertility and baby maize yield. Nitrogen 2026;7(1): 32.
43. Chuong NV. Response of peanut quality and yield to chicken manure combined with Rhizobium inoculation in sandy soil. Commun Sci Technol. 2023;8:31-7. [CrossRef]
44. Peix A, Ramírez-Bahena MH, Velázquez E, Bedmar E. Bacterial associations with legumes. Crit Rev Plant Sci. 2015;34:17-42. [CrossRef]
45. López-Fernández S, Mazzoni V, Pedrazzoli F, Pertot I, Campisano A. A phloem-feeding insect transfers bacterial endophytic communities between grapevine plants. Front Microbiol. 2017;8:834. [CrossRef]
46. Cardoso P, Alves A, Silveira P, SáC, Fidalgo C, Figueira E. Bacteria from nodules of wild legume species:Phylogenetic diversity, plant growth promotion abilities and osmotolerance. Sci Total Environ. 2018;645:1094-92. [CrossRef]
47. Etesami H. Root nodules of legumes:A suitable ecological niche for isolating non-rhizobial bacteria with biotechnological potential in agriculture. Curr Res Biotechnol. 2022;4:78-86. [CrossRef]
48. Dutta P, Ghosh M. Plant growth-promoting rhizobacteria (PGPR) and its mechanisms against plant diseases for sustainable agriculture and better productivity. BIOCELL. 2022;46(8):1843-59. [CrossRef]
49. Vurukonda SS, Giovanardi D, Stefani E. Plant growth promoting and biocontrol activity of Streptomyces spp. as endophytes. Int J Mol Sci. 2018;22(4):952. [CrossRef]
50. Trivedi T, Leach JE, Tringe SG, Sa T, Singh BK. Plant-microbiome interactions:From community assembly to plant health. Nat Rev Microbiol. 2020;18(11):607-21. [CrossRef]
51. Ahmad HM, Fiaz S, Hafeez S, Zahra S, Shah AN, Gul B, et al. Plant growth-promoting rhizobacteria eliminate the effect of drought stress in plants:A review. Front Microbiol. 2022;13:875774. [CrossRef]
52. Yang P, Condrich A, Scranton S, Hebner C, Lu L, Ali MA. Utilizing plant growth-promoting rhizobacteria (PGPR) to advance sustainable agriculture. Bacteria. 2024;3(4):434-51. [CrossRef]
53. Nguyen VC, Trang NP, Liem TT, Dang PT. Effect of Bacillus sonklengsis associated with cattle manure fertilization on the farmland health and peanut yield. Int J Agric Biosci. 2025;14(3):496-503. [CrossRef]
54. Van NC. The effectiveness of chemical fertilizer combined with lime, cow manure and indigenous nitrogen-fixing bacteria inoculation on soil fertility and white bean yield. Malays J Soil Sci. 2025;29:19-28.
55. Nguyen VC, Tri TL, Tuan LM. Assessing the superiority of Bacillus songklensis strain kca6 along with lime and cow manure to increase white bean yield in cadmium contaminated soil. Aust J Crop Sci. 2024;18(11):768-774.[CrossRef]
56. Maroniche GA, Puente ML, García JE, Mongiardini E, Coniglio A, Nievas S, et al. Phenogenetic profile and agronomic contribution of Azospirillum argentinense Az39T, a reference strain for the South American inoculant industry. Microbiol Res. 2024;283:127650. [CrossRef]
57. Pratap V, Dass A, Dhar S, Kumari K, Sudhishri S. Precision nitrogen, irrigation and cultivation regimes for enhanced yield, and nutrient accumulation in direct-seeded basmati rice (Oryza sativa). Indian J Agric Sci. 2024;94(6):571-6. [CrossRef]
58. Kumar BP, Trimurtulu N, Gopal AV, Nagaraju Y. Impact of culturable endophytic bacteria on soil aggregate formation and peanut (Arachis hypogaea l.) growth and yield under drought conditions. Curr Microbiol. 2022;79:308. [CrossRef]
59. Zhang T, Jian Q, Yao X, Guan L, Li L, Liu F, et al. Plant growth-promoting rhizobacteria (PGPR) improve the growth and quality of several crops. Heliyon. 2024;10(10):e31553. [CrossRef]
60. Haskett TL, Tkacz A, Poole PS. Engineering rhizobacteria for sustainable agriculture. ISME J. 2021;15:949-64. [CrossRef]
61. Li Y, Narayanan M, Shi X, Chen X, Li Z, Ma Y. Biofilms formation in plant growth-promoting bacteria for alleviating agro-environmental stress. Sci Total Environ. 2024;907:167774. [CrossRef]
62. Nedumaran S, Sharma DK, Bhatia A, Shrivastava M, Shivay YS, Mohan D, et al. Interactive effect of ambient and elevated levels of tropospheric ozone, nutrition and PGPR on growth and yield of chickpea (Cicer arietinum). Indian J Agric Sci. 2024;94(5):507-11. [CrossRef]
63. De Andrade LA, Santos CH, Frezarin ET, Sales LR, Rigobelo EC. Plant growth-promoting Rhizobacteria for sustainable agricultural production. Microorganisms. 2023;11(4):1088. [CrossRef]
64. Ballat EM, Elsilk SE, Ali HM, Ali HE, Hano C, El-Esawi MA. Metal-resistant PGPR strain Azospirillum brasilense EMCC1454 enhances growth and chromium stress tolerance of chickpea (Cicer arietinum L.) by modulating redox potential, osmolytes, antioxidants, and stress-related gene expression. Plants (Basel). 2023;12:2110. [CrossRef]
65. Padbhushan R, Sinha AK, Kumar U, Bhattacharya PM, Poddar P. Plant Growth-Promoting Bacteria and Crop Residue in Rice–Wheat System Cultivated with Favorable Tillage Influence Crop Productivity, Nutrient Uptake, Soil Quality, and Profitability in the Terai Agro-Ecological Zone of West Bengal, India. Agronomy. 2023;13(10):2454. [CrossRef]
66. Motlagh MR, Farokhzad M, Kaviani B, Kulus D. Endophytic fungi as potential biocontrol agents against Sclerotium rolfsii Sacc. the causal agent of Peanut white stem rot disease. Cells. 2022;11:2643. [CrossRef]