1. INTRODUCTION
Cotton plantations have long been a cornerstone of global agriculture, contributing significantly to economies and trade networks across the world. However, the challenges posed by pests to these plantations have a substantial impact on both crop yields and the broader economy [1]. Recent data highlights the complex interplay between cotton pests and economic consequences, shedding light on the urgent need for innovative pest management strategies to secure sustainable cotton production and maintain stable economies [2]. Recent data underscores the severity of the economic impact. According to the Mississippi Agricultural and Forestry Experiment Station, in 2022, the loss caused by pests in cotton production in the United States, one of the largest producers worldwide, was equal to 4.80%, generating a sum of cost and economic loss equal to $ 777,281,062 dollars. Such losses ripple through various sectors, from textile manufacturing and trade to employment and local economies [3].
In addition to direct yield losses, pest-related challenges also escalate production costs. Farmers often resort to increased pesticide applications to curb pest infestations, contributing to rising expenses and potentially harmful environmental consequences. Furthermore, the emergence of pesticide-resistant pest populations compounds the challenge, necessitating further investments in research and pest management strategies to maintain effective control [4]. Cotton cultivation, particularly in regions heavily reliant on this cash crop, forms the backbone of local economies. Recent data from developing countries such as India, China, and parts of Africa illustrate the intricate link between cotton production and economic stability. For instance, a study conducted by the Indian Council for Research on International Economic Relations revealed that cotton accounts for over 5% of India’s total agricultural output and directly supports millions of smallholder farmers [5]. The economic dynamics of cotton plantations extend beyond the farm gate. Textile industries, processing mills, and export markets all depend on a consistent and quality supply of cotton. Pest-related disruptions can lead to reduced availability, higher prices, and decreased competitiveness in international markets, affecting trade balances and overall economic growth [6].
The genus Dysdercus comprises a group of hemipteran insects known for their significant impact as pests in cotton cultivation, which includes Dysdercus peruvianus (Figure 2), Dysdercus koenigii and Dysdercus cingulatus, for example. These insects, commonly referred to as cotton stainers, are notorious for causing substantial economic losses by inflicting damage to cotton crops [1,7,8]. Dysdercus species feed on various plant parts, including developing bolls, seeds, and leaves, resulting in reduced yields and compromised fiber quality. The staining caused by their feeding renders cotton fibers unsuitable for textile processing, leading to additional financial setbacks for cotton farmers and industries [9]. Integrated pest management strategies, research on pest biology, and the development of resistant cotton varieties are essential components of efforts to mitigate the damaging effects of Dysdercus species on cotton production and uphold the economic viability of this crucial agricultural sector [10,11].
 | Figure 1: Example of plants with high activity essential oils against Dysdercus spp. From left to right: Pilocarpus spicatus (Source: Milton Groppo); Boswellia serrata (Dinesh Valke); Ocimum sanctum. [Click here to view] |
 | Figure 2: Dysdercus peruvianus adult (Source: Rob Westerduijn). [Click here to view] |
The control of Dysdercus species, notorious cotton pests, has historically relied heavily on synthetic pesticides, aiming to mitigate the economic losses inflicted on cotton crops. However, the prolonged and indiscriminate use of these pesticides has led to a host of environmental and ecological problems, including the development of pesticide-resistant pest populations and harm to non-target organisms [12]. In response, the search for sustainable alternatives has gained momentum, with natural products, including essential oils (EOs), emerging as promising candidates [13,14]. EOs with high yields are considered effective and low-cost resources, in addition to being eco-friendly and less susceptible to the development of resistance by pests [15]. In addition to insecticidal use, they can also act as antibacterial and antifungal agents of agricultural and food importance [16-18] and can be applied through suitable methods according to the need, which include spray and mixing with soil components [19,20]. Furthermore, recent biotechnological resources such as the development of nanostructured systems, for example, nanoemulsions and nanocapsules, are also considered promising for the inclusion of EOs as active agents [21,22]. Regarding insecticidal activity, its various components, mainly oxygenated monoterpenes, and phenylpropanoids, can act through different mechanisms of action, such as, for example, inhibition of acetylcholinesterase, activation of chlorine channels, as well as genotoxic and antifeedant effects [23].
EOs derived from various plant sources possesses insecticidal properties that can effectively target Dysdercus pests while minimizing negative ecological impacts. These natural products offer a sustainable alternative to synthetic pesticides, reducing the risk of resistance development and preserving beneficial organisms within the ecosystem. Moreover, EOs have shown potential in integrated pest management strategies, promoting a holistic approach to pest control that aligns with modern agricultural sustainability goals [24]. As research continues to advance, the integration of natural products like EOs into cotton pest management practices holds promise for maintaining crop productivity while fostering environmentally responsible and resilient agricultural systems.
2. METHODOLOGY
The research was carried out using the combination of “essential oil” and “Dysdercus”. The following research databases were used to the review: Science Direct (56 documents), Scopus (264 documents), and Google Scholar (905 documents). According to the relevance order, repeated articles or those that only cited studies on the subject in question were excluded, so that 98 articles had their content analyzed and only studies within the central theme were selected. Then, after careful selection, 19 articles were included and discussed in the two next sections of the review, which excludes the articles described in the introduction and discussion sections. Table 1 summarizes the selected articles and their main information.
Table 1: Essential oils with activity on Dysdercus spp.
| Affected pest | Plant species | Family | Drug plant | Main constituents | Biological activity | References |
|---|---|---|---|---|---|---|
| Dysdercus peruvianus | Persea venosa | Lauraceae | Leaves | β-caryophyllene (43.78%), caryophyllene oxide (8.92%), α-humulene (8.27%), and α-copaene (5.90%) | LC50=24.45 μg/µL (oil), LC50=28.73 μg/µL (nanoemulsion) against 4th instar nymphae, after 20 days | [25] |
| Pilocarpus spicatus | Rutaceae | Sabinene (32.27%) and sylvestrene (27.26%) | LC50=90 μg/mL (topical), LC50=110.88 μg/cm2(direct contact) against 5th instar nymphae, after 22 days; insect malformation | [26] | ||
| Ocotea indecora | Lauraceae | Sesquirosefuran (92%) | LC50=162.18 μg/insect (oil), 5% nanoemulsion caused 90% mortality against 4th instar nymphae, after 20 days; IC50=1.37 mg/mL (oil) on acetylcholinesterase enzyme | [27] | ||
| 62.5 mg/mL oil caused 100 % mortality against 4th instar nymphae, after 24 h; insect malformations | [9] | |||||
| Myrtaceae spp. | Myrtaceae | NI | pure oil caused 100% mortality against 4th instar nymphae, after 30 days; insect malformations | |||
| Zanthoxylum caribaeum | Rutaceae | Sylvestrene (11.3%), muurola-4 (14),5-trans-diene (8.4%), isodaucene (8.3%) and α-Pinene (7.6%) | 500 μg/insect oil caused 100 % mortality against 4th instar nymphae, after 24 h; insect malformations and development interruption | [28] | ||
| Myrciaria floribunda | Myrtaceae | 1,8-cineole (38.4%) | LC50 = 309.64 μg/insect against adults, after 22 days | [29] | ||
| Eugenia sulcata | β-caryophyllene (24.6 %), α-pinene (17.2%) and β-pinene (10.9%) | 500 mg/mL oil caused 66.7 % mortality against 4th instar nymphae, after 24 h | [30] | |||
| Xylopia ochrantha | Annonaceae | Germacrene D (17.8%) and bicyclogermacrene (17.4%) | pure oil caused 88% mortality in 24 h and 100% in 10 days; 5% nanoemulsion caused 18% mortality in 24 h and 100% in 10 days against 4th instar nymphae; insect malformations | [31] | ||
| Cymbopogum winterianus | Poaceae | caryophyllene (26.50%) and citronellal (26.02%) | pure oil caused 100% mortality in 24 h against 4th instar nymphae; insect malformations | |||
| Dysdercus koenigii | Artemisia annua | Asteraceae | Aerial parts | NI | LC50=0.48 μL against 5th instar nymphae, after 48 h; poor ovary development; greater median neurosecretory activity | [32] |
| Callistemon lanceolatus | Myrtaceae | NI | Eugenol (NI) | Inhibition of development in 5th instar larvae | [33] | |
| Eucalpytus globulus | Leaves | NI | 35.7% mortality occurred in 15-day old nymphs, exposed during 5 h exposure to oil vapour; weight loss, inhbition of fecundity and egg hatching | [34] | ||
| Acorus calamus | Acoraceae | Inhibition of male fecundity (20 μL: Total inhibition) | [35] | |||
| Inhibition of female fecundity (100 μL: Total inhibition) | [36] | |||||
| Inhibition of egg hatching and nymphae development; 100% mortality of survivors in 24 h (5 µL) | [37] | |||||
| Dysdercus similis | Boswellia serrata | Burseraceae | Gum resin | Myrcene, D-α-caryophyllene, β-caryophyllene, limonene, dipentene, α-terpinene, p-cymene, terpinen-4-ol, α-pinene, β-pinene, α-thujene, camphene and bornyl acetate | 1:30 dilution caused histological derangements on male gonads of 5th instar nymphs | [38] |
| α-pinene (40.32%), β-pinene (10.43%) | Pure oil and 1:5 dilution caused 100% mortality; 1:10 to 1:50 caused juvenomimetic effect (93.75 to 61.25%) | [39] | ||||
| Dysdercus voelkeri | Ocimum sanctum | Lamiaceae | Dry inflorescences | Germacrene D (25%), β-caryophyllene (21.28%), methyl-eugenol (14.25%), eugenol (10.78%), β-elemene (9.78%) and elemol (7.64%) | 1 µl/L concentration caused 100% mortality on three different development nymph stages (II, III and IV) within 24 h; LC50=between 0.536 and 0.590 µl/L; LC50 for adults=0.627 µl/L. | [40] |
| Cymbopogon schoenanthus | Poaceae | Leaves | Piperitone (69,80%) and δ-2-carene (18,48%) | 6% concentration (1.8 L/ha) caused 71.40% mortality on adult insects. | [41] | |
| Dysdercus cingulatus | Acorus calamus | Acoraceae | NI | Low activity on the egg hatching (circa 15%) and moderate activity adult emergence from eggs and nymphae (circa 50%), using the range dose between 2 and 4 μL, after 24 h of paper filter contact treatment. The 1 μL dose caused circa 25% inhibition of adult emergence, whereas the 3 μL dose led to circa 33 and 43% inhibition for nymphae and egg emergence, respectively | [42] |
3. EOs WITH ACTIVITY ON DYSDERCUS GENUS
3.1. D. peruvianus
Esteves et al. (2023) conducted a study in which they examined the insecticidal properties of Persea venosa leaf EO on 4th instar nymphs. The results revealed an LC50 of 24.45 μg/µL for the pure EO, while the nanoemulsified EO exhibited an LC50 of 28.73 μg/µL. This assessment involved a 20-day topical treatment. Notably, the pure oil, particularly at a concentration of 450 μg/µL, and its primary component, β-caryophyllene, led to 100% mortality within the 1st day. In addition, it’s worth mentioning that the EO showed no toxicity towards Apis mellifera and Partamona helleri, underscoring its selectivity. The major compounds identified were β-caryophyllene (43.78%), caryophyllene oxide (8.92%), α-humulene (8.27%), and α-copaene (5.90%) [25]. A similar investigation by Apolinário et al. (2020) focused on the EO extracted from Pilocarpus spicata leaves and its impact on 5th instar nymphs. After 22 days of continuous treatment, the LC50 values for topical treatment and paper filter contact were found to be 90 μg/mL and 110.88 μg/cm2, respectively. Furthermore, the treatment often resulted in insect malformations and the production of permanent or supernumerary nymphs. The major constituents of the oil were sabinene (32.27%) and sylvestrene (27.26%) [26]. Ocotea indecora leaf EO demonstrated significant insecticidal activity against 4th instar nymphs, with an LC50 value of 162.18 μg/insect. The nanoemulsified version, with a 5% oil concentration, induced 90% mortality after 20 days of treatment. Notably, surviving insects proceeded to subsequent instars and adulthood, while deceased insects exhibited characteristic symptoms of anticholinesterase toxicity, as further assessed with an IC50 of 1.37 mg/mL on the acetylcholinesterase enzyme. The primary compound identified was sesquirosefuran (92%) [27]. Costa (2023) explored the insecticidal effects of EOs from two species: O. indecora and an unidentified Myrtaceae species. The O. indecora oil achieved 100% mortality among 4th instar nymphs within 24 h at a concentration of 62.5 mg/mL. In contrast, the Myrtaceae oil achieved the same mortality rate only after 30 days of topical treatment with the pure oil, with no fatalities within the first 24 h. Both oils interfered with nymph metamorphosis and caused deformations in the insect cuticle structure. Sesquirosefuran (92.2%) was the major compound in O. indecora EO [9]. The insecticidal activity of Zanthoxylum caribaeum leaf EO was also evaluated on 4th instar nymphs of D. peruvianus. The pure oil and a dose of 500 μg/insect induced 100% mortality within 24 h, with an LD50 of 215 μg/insect after 2 days. The oil also disrupted metamorphosis and molting, often in a dose-dependent manner, leading to nymphs with deformed legs, wings, and antennae. Major compounds identified included sylvestrene (11.3%), muurola-4(14),5-trans-diene (8.4%), isodaucene (8.3%), and α-pinene (7.6%) [28]. Another EO, derived from the Brazilian sandbank plant Myrciaria floribunda, exhibited insecticidal effects against D. peruvianus adults. It displayed an LD50 equal to 309.64 µg/insect after 24 h and 94.42 µg/insect after 22 days of treatment. The primary constituent of this oil was 1,8-cineole (38.4%) [29]. Eugenia sulcata leaf EO, at a concentration of 500 mg/mL, caused 66.7% mortality in 4th instar nymphs within 24 h and 80% after 10 days of treatment using the same direct contact model. β-caryophyllene (24.6%) was the predominant constituent, followed by α-pinene and β-pinene at 17.2% and 10.9%, respectively [30]. Finally, Nunes (2022) explored the insecticidal properties of two additional EOs, specifically from Xylopia ochrantha and Cymbopogon winterianus, on 4th instar nymphs. X. ochrantha EO induced 88% mortality within 24 h for the pure oil and 100% within 10 days. In contrast, the nanoemulsion of the same oil (5%) led to 18% mortality in 24 h and 100% within 10 days. Meanwhile, C. winterianus pure EO resulted in 100% insect mortality within 24 h, and a concentration of 500 µg/insect caused 86% mortality in the same period. Both EOs affected nymph development inhibited egg viability and led to structural malformations. The major compounds identified were germacrene D (17.8%) and Bicyclogermacrene (17.4%) in X. ochranta oil, and caryophyllene (26.50%) and citronellal (26.02%) in C. winterianus oil [31].
3.2. D. koenigii
Artemisia annua oil exhibited potent insecticidal activity and had notable effects on the development and reproduction of D. koenigii. The LD50 for 5th instar nymphs was determined to be 0.48 μL, 48 h after topical treatment. The surviving nymphs displayed a delayed development of up to 2 days, resulting in intermediates. In addition, the treatment led to a decrease in hemolymph protein concentration and disrupted its electrophoretic protein pattern. Furthermore, adults derived from treated nymphs exhibited underdeveloped ovaries, and there was an observed increase in median neurosecretory activity in the treated insects for up to 6 days into adulthood [32]. Another significant finding was the juvenoid effect of Callistemon lanceolatus EO, rich in eugenol, on D. koenigii. This effect was observed when the oil was topically applied to the last instar larva [33]. Furthermore, Srivastava et al. (1995) conducted a study evaluating the impact of eucalyptus EO vapors on nymphs aged 3–15 days, with exposure lasting from 2 to 5 h. The highest mortality rate (35.7%) occurred among 15-day-old nymphs after their fifth exposure. In addition, there was a significant reduction in the total postembryonic developmental period of the surviving nymphs, particularly when 10- and 15-day-old nymphs were exposed for 4 or 5 h, respectively. Surviving adults who emerged from this exposure exhibited weight loss, reduced fecundity, and decreased egg hatchability [34]. Furthermore, it was reported that the vapors from Acorus calamus leaf EO had a significant downregulating effect on the fecundity of D. koenigii males (total inhibition at 20 μL) and females (total inhibition at 100 μL). In addition, these vapors strongly inhibited egg hatching and nymph development, resulting in the mortality of all survivors, even when the lower dose of 5 μL was used, all within a 24-h period [35-37].
3.3. D. similis
In the study conducted by Rao and Kaur (1989), they investigated the impact of topically applying a Boswellia serrata (Figure 1) gum resin-derived EO on the male gonads of 5th instar nymphs. The EO was administered using a dilution of 1:30 in acetone. Notable histological derangements were observed in the male gonads as a result of this treatment. The EO was found to contain various compounds, including myrcene, D-α-caryophyllene, β-caryophyllene, limonene, dipentene, α-terpinene, p-cymene, terpinen-4-ol, α-pinene, β-pinene, α-thujene, camphene, and bornyl acetate [38]. In addition, a similar study revealed the juvenomimetic activity of the same EO on these nymphs. The percentage of juveniles varied from 93.75% to 61.25%, depending on the concentration range used, which ranged from 1:10 to 1:50 in acetone. Notably, the pure oil and the 1:5 dilution caused 100% mortality. The primary constituents of the EO were identified as α-pinene (40.32%) and β-pinene (10.43%) [39].
3.4. D. voelkeri
The EO from Ocimum sanctum dry inflorescences (Figure 1) caused 100% mortality in three different development nymph stages (II, III, and IV) within 24 h, using a 1 µL/L concentration. The LC50 value found was in a range between 0.536 and 0.590 µL/L, being similar to the positive control. On the other side, the LC50 value for adults was 0.627 µl/L. Germacrene D (25%), β-caryophyllene (21.28%), methyl-eugenol (14.25%), eugenol (10.78%), β-elemene (9.78%), and elemol (7.64%) were the major compounds of the oil [40]. Part of the previous researchers group also showed that the oil/water emulsion of Cymbopogon schoenanthus EO (6%; 1.8 L/ha) caused 71.40% mortality in adult insects. Piperitone (69.80%) and δ-2-carène (18.48%) were the major compounds of the oil [41].
3.5. D. cingulatus
D. cingulatus is an important cotton pest in Pakistan and attacks other species of Malvaceae family. In a study conducted by Baloch (1990), it was described that the EO vapors from A. calamus, a national plant species, demonstrated low activity on the egg hatching (circa 15%) and moderate activity on adult emergence from eggs and nymphae (circa 50%), using the range dose between 2 and 4 μL, after 24 h of paper filter contact treatment. Moreover, the 1 μL dose caused circa 25% inhibition of adult emergence, whereas the 3 μL dose led to circa 33 and 43% inhibition for nymphaea and egg emergence, respectively [42].
4. DISCUSSION
Overall, 19 documents were found on the insecticidal activity of EOs against insects of the genus Dysdercus. Among the indicated studies, most declared the action of EOs from plants found in the sandbank vegetation of the Brazilian coast against the species D. peruvianus. The insecticidal activity of these plants has been extended studied against agricultural pests and disease transmitters [43]. Especially, the EO from P. spicatus leaves showed marked activity against 5th stage nymphs, with an LC50 equal to 90 μg/mL, in the topical assay. The hydrocarbon monoterpenes sabinene and sylvestrene were the major compounds of the EO. Sabinene proved to be a substance that can act as a synergistic promoter of other active components in the EO, such as 4-terpineol, also present in this oil, with a composition above 2% of the total identified [26,44]. Furthermore, the hydrocarbon monoterpenes also have nonspecific mechanisms of action such as disruption of the cellular membrane, which can lead to damage to the tissues that are more easily exposed [45].
In addition, the other mentioned EOs also had relevant activity against adult insects or nymphs, so other important data, such as the absence of toxicity in non-target organisms observed for P. venosa and the effectiveness of innovative formulations such as nanoemulsions, are important factors to be considered for EOs that did not show activity as high as P. spicatus. Nanoemulsions are formulations that have advantages such as better dispersion of oils in an aqueous environment, such as greater stability of actives and increased bioavailability [46].
It also highlighted the insecticidal activities of A. annua and B. serrata oils against 5th instar nymphs of D. koenigii and D. similis, respectively, while the EO from O. sanctum dry inflorescences showed high efficiency against three stages of D. voelkeri nymphs. The composition of this oil is rich in sesquiterpenes, phenylpropanoids, and oxygenated monoterpenes. The insecticidal activity of β-caryophyllene was already seen against different families such as Spodoptera frugiperda, Myzus persicae and Aphis gossypii, so that it caused significant adverse effects on the fertility, as well as on the honeydew excretion frequency and production. Furthermore, β-caryophyllene inhibited the activities of esterases, oxidases, and glutathione S-transferases [47-49]. Among the main phenylpropanoids (aromatic terpenes) are found the eugenol and methyl eugenol, which in turn possess relevant insecticidal activities against different pests [50,51]. Moreover, aromatic terpenes can act through different mechanisms, including ligand-gated chloride channels enhancing in insect nervous system and octopamine receptors blocking [52]. In addition, oxygenated monoterpenes, as the linalool, are related to higher biological activity in the insect nervous system, due to the presence of a strong electronegative heteroatom (oxygen), which causes specific interactions with enzymes and receptors [53]. On the other hand, the demonstration of insecticidal activity in the field was also important, as described for the oil of C. schoenanthus against D. voelkeri. This study validates the practical performance of the use of this product as an insecticide, which, combined with the economic practicality and ease of oil obtaining, presents itself as a real candidate for a natural biological regulator.
In addition to all the factors discussed above, effects on development, metamorphosis, feeding, and laying/hatching of eggs must also be taken into account. Since insect mortality is not always fully achieved, these effects are important for pest control, as they prevent the healthy development or reproduction of these insects, reducing the loss of agricultural products [54,55].
In conclusion, the presented and discussed studies were channeled in this review towards an organized and integrated view of their meanings, to inform the importance of EOs as potential biocontrollers of different Dysdercus species (Figure 3), so that combined with the biotechnological advances cited in some studies, such as incorporation into nanostructured systems for example nanoemulsions, can promote the use of more sustainable and efficient methods for controlling pests that affect cotton production in several countries around the world, so that Despite these findings, further detailed studies on chemical standardization of EOs, development of suitable formulations and cost-efficiency evaluation must be carried out.
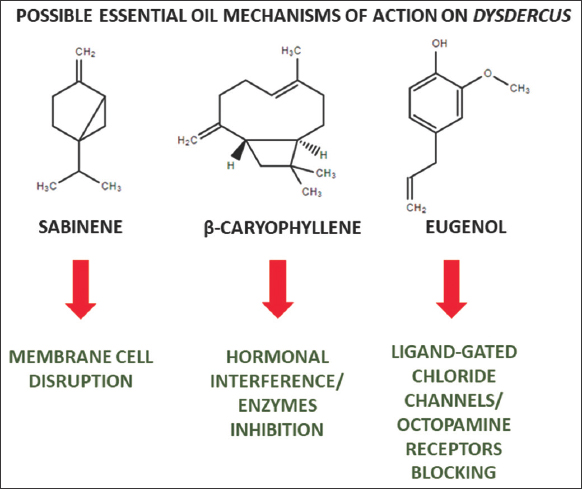 | Figure 3: Some essential oils major constituents and their probable mechanisms of action on Dysdercus insects. [Click here to view] |
5. CONCLUSION
The review highlights the role of EOs as promising biocontrol agents against Dysdercus insects in cotton plantations. Diverse oils, notably P. spicatus, A. annua, B. serrata, and O. sanctum, exhibit relevant insecticidal activity due to compounds such as monoterpenes and phenylpropanoids, which in turn have noted insecticide mechanisms, such as membrane cell disruption, hormonal interference, and nervous system toxicity. Moreover, practical field applications and consideration of developmental effects on pests further emphasize the potential of EOs for sustainable pest management, offering a viable and sustainable solution for global cotton production challenges.
6. AUTHOR CONTRIBUTIONS
All authors made substantial contributions to the conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
7. FUNDING
There is no funding for this article.
8. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
9. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
10. DATA AVAILABILITY
All the data is available with the authors and shall be provided upon request.
10. USE OF ARTIFICIAL INTELLIGENCE (AI)-ASSISTED TECHNOLOGY
The authors declares that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
11. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Razaq M, Mensah R, Athar HU. Insect pest management in cotton. In:Cotton Production. Hoboken:John Wiley and Sons Ltd.;2019. 85-107.
2. Chi BJ, Zhang DM, Dong HZ. Control of cotton pests and diseases by intercropping:A review. J Integr Agric 2021;20:3089-100. [CrossRef]
3. MSU (Mississippi Agricultural and Forestry Experiment Station). Cotton Insect Losses;2022. Available from:https://www. biochemistry.msstate.edu/resources/pdf/2022/table31.pdf. [Last accessed on 2023 Dec 10].
4. Jabran K, Ul-Allah S, Chauhan BS, Bakhsh A. An introduction to global production trends and uses, history and evolution, and genetic and biotechnological improvements in cotton. In:Cotton Production. Hoboken:John Wiley and Sons Ltd.;2019. 1-22.
5. Wardhan H. Agricultural Value Chains in India:Ensuring Competitiveness, Inclusiveness, Sustainability, Scalability, and Improved Finance. Germany:Springer;2022. 4-281.
6. Khan MA, Wahid A, Ahmad M, Tahir MT, Ahmed M, Ahmad S, et al. World cotton production and consumption:An overview. In:Cotton Production and Uses:Agronomy, Crop Protection, and Postharvest Technologies. Singapore:Springer;2020a. 1-7.
7. Karar H, Bashir MA, Haider M, Haider N, Hassan M, HashemM, et al. Ecological impact on development of hemipterous bug (Dysdercus koenigii) (Hemiptera:Pyrrhocoridae) and boll rot disease of cotton (Gossypium hirsutum) grown in the diversified field. Saudi J Biol Sci 2021;28:3957-64. [CrossRef]
8. Khan WA, Qamar SU, Ahmad JN, Calma ML, Ullah A. Phylogenetic analysis of red cotton bug species (Hemiptera:Pyrrhocoridae) in Punjab, Pakistan. Acta Entomol Serbica 2020b;25:1-11.
9. Costa MG. Estudo da Atividade dos Óleos Essenciais de Ocotea elegans (Lauraceae) e de uma Myrtaceae nos Hemiptera Oncopeltus fasciats (Lygaeidae) e Dysdercus peruvianus (Pyrrhocoridae). Master Thesis. Universidade Federal Fluminense;2023.
10. Hussain M, Kacho NF, Mohan S, Mir AH. Potential control agents against red cotton bug Dysdercus koenigii-a review. Indian J Entomol 2021;83:1-8. [CrossRef] [CrossRef] [CrossRef]
11. Montero-Peña JM. Aplicación del Espectro Visible Para el Control de la Plaga del Arrebiatado (Dysdercus peruvianus Guerin) en el Cultivo de Algodón-Piura. PhD Thesis. Universidad Nacional de Piura;2021.
12. Saeed R, Abbas N, Hafez AM. Biological fitness costs in emamectin benzoate-resistant strains of Dysdercus koenigii. Entomol Gen 2021;41:267-78. [CrossRef]
13. Singh-Gupta S, Magdum S, Jawale CS. Acute toxicity of menadione on DNA content in the gonads of Dysdercus cingulatus. Trends Life Sci 2020;9:1-4.
14. Shahzad A, Khan MA, Ashfaq M, Ahmed JN, Zahid M, Mustafa FU, et al. Comparative effect of botanical extracts and synthetic pesticides against Dysdercus cingulatus F. and Amrasca biguttula I. in Okra. J Entomol Zool Stud 2019;7:1291-5.
15. Chaudhari AK, Singh VK, Kedia A, Das S, Dubey NK. Essential oils and their bioactive compounds as eco-friendly novel green pesticides for management of storage insect pests:Prospects and retrospects. Environ Sci Pollut Res Int 2021;28:18918-40. [CrossRef]
16. Wang J, Zhao F, Huang J, Li Q, Yang Q, Ju J. Application of essential oils as slow-release antimicrobial agents in food preservation:Preparation strategies, release mechanisms and application cases. Crit Rev Food Sci Nutr 2023; 18:1-26.
17. Wu H, Zhao F, Li Q, Huang J, Ju J. Antifungal mechanism of essential oil against foodborne fungi and its application in the preservation of baked food. Crit Rev Food Sci Nutr 2022;120:1-13.
18. Ju J, Guo Y, Cheng Y, Yaoc W. Analysis of the synergistic antifungal mechanism of small molecular combinations of essential oils at the molecular level. Ind Crops Prod 2022;188:115612. [CrossRef]
19. Isman MB. Commercial development of plant essential oils and their constituents as active ingredients in bioinsecticides. Phytochem Rev 2020;19:235-41. [CrossRef]
20. Shaltiel-Harpaz L, Kreimer T, Dudai N, Kaspi R, Ben-Yakir D, Rytwo G. Sepiolite-rosemary oil combination as an environmentally oriented insecticide. Appl Clay Sci 2023;234:106838. [CrossRef]
21. Singh IR, Pulikkal AK. Preparation, stability and biological activity of essential oil-based nano emulsions:A comprehensive review. OpenNano 2022;8:100066. [CrossRef]
22. Khoobdel M, Ahsaei SM, Farzaneh M. Insecticidal activity of polycaprolactone nanocapsules loaded with Rosmarinus officinalis essential oil in Tribolium castaneum (Herbst). Entomol Res 2017;47:175-84. [CrossRef]
23. Arshad Z, Hanif MA, Qadri RW, Khan MM, Babarinde A, Omisore GO, et al. Role of essential oils in plant diseases protection:A review. Int J Chem Biochem Sci 2014;6:11-7.
24. Gupta I, Singh R, Muthusamy S, Sharma M, Grewal K, Singh HP, et al. Plant essential oils as biopesticides:Applications, mechanisms, innovations, and constraints. Plants (Basel) 2023;12:2916.
25. Esteves RS, Apolinário R, Machado FP, Folly D, Viana VC, Soares AP, et al. Insecticidal activity evaluation of Persea venosa Nees &Mart. essential oil and its nanoemulsion against the cotton stainer bug Dysdercus peruvianus (Hemiptera:Pyrrhocoridae) and pollinator bees. Ind Crops Prod 2023;194:116348. [CrossRef]
26. Apolinário R, Nogueira J, da Silveira Costa MG, Santos-Mallet J, Santos MG, Azambuja P, et al. Insecticidal activity of Pilocarpus spicatus Saint-Hilaire (Rutaceae) essential oil against the crop pest Dysdercus peruvianus (Guérin-Méneville, 1831) and Oncopeltus fasciatus (Dallas, 1852). Res Soc Dev 2020;9:e90091110489.
27. Nascimento LM, Apolinario R, Machado FP, Correa AL, Caldas GR, Ruppelt BM, et al. Effects of nanoemulsion and essential oil from the leaves of Ocotea elegans against Dysdercus peruvianus. Res Soc Dev 2020;9:e909108424.
28. Pacheco JP, Nogueira J, de Miranda RP, Duprat RC, Machado FP, Tietbohl LA, et al. Effects of Zanthoxylum caribaeum essential oil against cotton bug Dysdercus peruvianus. Res Soc Dev 2020;9:e197997152.
29. Tietbohl LA, Barbosa T, Fernandes CP, Santos MG, Machado FP, Santos KT, et al. Laboratory evaluation of the effects of essential oil of Myrciaria floribunda leaves on the development of Dysdercus peruvianus and Oncopeltus fasciatus. Rev Bras Farmacogn 2014;24:316-21. [CrossRef]
30. Gonzalez MS, Lima BG, Oliveira AF, Nunes DD, Fernandes CP, Santos MG, et al. Effects of essential oil from leaves of Eugenia sulcata on the development of agricultural pest insects. Rev Bras Farmacogn 2014;24:413-8. [CrossRef]
31. Nunes DD. Potencial do Óleo Essencial de Xylopia ochrantha Mart. E Cymbopogon winterianus Jowitt. Ex Bor Como Inseticidas Botânicos no Controle de Dysdercus peruvianus (Guérin-Méneville, 1831) e Oncopeltus fasciatus (Dallas, 1852). PhD Thesis. Universidade Federal Fluminense;2023.
32. Rao PJ, Kumar KM, Singh S, Subrahmanyam B. Effect of Artemisia annua oil on development and reproduction of Dysdercus koenigii F.(Hem., Pyrrhocoridae). J Appl Entomol 1999;123:315-8. [CrossRef]
33. Katiyar RL, Srivastava KP. An essential oil from Australian bottlebrush, Callistemon lanceolatus (Myrtaceae) with juvenoid properties against the red cotton bug, Dysdercus koenigii Fabr. (Heteroptera, Pyrrhocoridae). Entomon 1982;7:463-8.
34. Srivastava RK, Krishna SS. Effect of exposure of eggs or first instar nymphs of Dysdercus koenigii (F.) (Heteroptera:Pyrrhocoridae) to eucalyptus oil odour on the insect's postembryonic development and/ or reproductive potential. Phytophaga 1995;4:47-52.
35. Saxena BP, Tikku K, Koul K. Spermatogenesis in Dysdercus koenigii and induced sterility by Acorus calamus oil vapours. Acta Entomol Bohemoslov 1977;74:381-7.
36. Saxena BP, Mathur AC. Loss of fecundity in Dysdercus koenigii F. due to vapours of Acorus calamus L. oil. Experientia 1976;32:313-6.
37. Saxena BP, Srivastava JB. Effect of Acorus calamus L. oil vapours on Dysdercus koenigii F. Indian J Exp Biol 1971;10:391-3.
38. Rao DR, Kaur A. Effect of the essential oil from the gum oleoresin of Boswellia serrata Roxb. On the gonads of male Dysdercus similis F. Curr Sci 1989;58:822-4.
39. Dennis TJ, Kumar KA, Srimannarayana G, Raghunathrao D. Juvenomimetic activity of the gumoleoresin of Boswellia serrata. Fitoterapia 1999;70:308-10. [CrossRef]
40. Nadio NA, Poutouli WP, Laba B, Tozoou P, Bokobana ME, Koba K, et al. Propriétés insecticides et répulsives de l'huile essentielle d'Ocimum sanctum L. envers Dysdercus voelkeri schmidt (Heteroptera; Pyrrhocoridae). Rev CAMES 2016;3:65-72.
41. Nadio NA, Bokobana EM, Akantetou KP, Tozoou P, Poutouli W, Koba K, et al. Efficacy of bioinsecticides based on the essential oil of Cymbopogon schoenanthus (L.) Spreng against red bugs (Dysdercus voelkeri Schmidt) in cotton cultivation in Togo Afr J Food Agric Nutr Dev 2021;21:17727-40.
42. Baloch AA. Effect of Acorus calamus L. oil vapours on egg development, fecundity and fertility of Dysdercus cingulatus Fabr. Ziraat Fak Derg 1990;6:213-24.
43. De Albuquerque RD, Rocha L. Essential oils from Carapebus sandbank plants as natural controllers against insect pests of importance in Brazilian public health and agriculture. Nat Resour Human Health 2023;3:315-22. [CrossRef]
44. Liu TT, Chao LK, Hong KS, Huang YJ, Yang TS. Composition and insecticidal activity of essential oil of Bacopa caroliniana and interactive effects of individual compounds on the activity. Insects 2019;11:23. [CrossRef]
45. Yap PS, Yusoff K, Lim SH, Chong CM, Lai KS. Membrane disruption properties of essential oils-a double-edged sword?Processes 2021;9:595.
46. Echeverría J, de Albuquerque RD. Nanoemulsions of essential oils:New tool for control of vector-borne diseases and in vitro effects on some parasitic agents. Medicines (Basel) 2019;6:42. [CrossRef]
47. Cárdenas-Ortega NC, González-Chávez MM, Figueroa-Brito R, Flores-Macías A, Romo-Asunción D, Martínez-González DE, et al. Composition of the essential oil of Salvia ballotiflora (Lamiaceae) and its insecticidal activity. Molecules 2015;20:8048-59. [CrossRef]
48. Chohan TA, Chohan TA, Mumtaz MZ, Alam MW, Naseer I, Riaz A, et al. Insecticidal potential of α-pinene and β-caryophyllene against Myzus persicae and their impacts on gene expression. Phyton 2023;92:1943-54. [CrossRef]
49. Liu YQ, Xue M, Zhang QC, Zhou FY, Wei JQ. Toxicity of β-caryophyllene from Vitex negundo (Lamiales:Verbenaceae) to Aphis gossypii glover (Homoptera:Aphididae) and its action mechanism. Acta Entomol Sin 2010;53:396-404.
50. Ulanowska M, Olas B. Biological properties and prospects for the application of eugenol-a review. Int J Mol Sci 2021;22:3671. [CrossRef]
51. Xu HX, Zheng XS, Yang YJ, Tian JC, Lu YH, Tan KH, et al. Methyl eugenol bioactivities as a new potential botanical insecticide against major insect pests and their natural enemies on rice (Oriza sativa). Crop Prot 2015;72:144-9. [CrossRef]
52. Tabari MA, Rostami A, Khodashenas A, Maggi F, Petrelli R, Giordani C, et al. Acaricidal activity, mode of action, and persistent efficacy of selected essential oils on the poultry red mite (Dermanyssus gallinae). Food Chem Toxicol 2020;138:111207. [CrossRef]
53. Moghaddam M, Mehdizadeh L. Chemistry of essential oils and factors influencing their constituents. In:Soft Chemistry and Food Fermentation. Cambridge:Academic Press;2017. 379-419.
54. Passos LC, Ricupero M, Gugliuzzo A, Soares MA, Desneux N, Campolo O, et al. Sublethal effects of plant essential oils toward the zoophytophagous mirid Nesidiocoris tenuis. J Pest Sci 2022;95:1609-19. [CrossRef]
55. Shilpa C, Remia KM. Bio-efficacy of microbial, chemical and conventional treatments against Spodoptera litura infesting gerbera plants. Memes Interdiscip Sci J 2017;1:56-67.