1. INTRODUCTION
Since the latter half of the 19th century, pathogenic fungi and bacteria have been known to generate chemical substances that cause symptoms to recur on test plants when the pathogen fails to thrive on it [1]. Nevertheless, in the middle of the 21st century, research on the toxins released by plant diseases and their impact on plants finally started to pick up steam [2,3]. The crucial role that toxins play in plant pathogenesis has long been a matter of debate. The comprehension of biological experiments intended for clarifying the role of pathogenic bacteria and fungi in disease production has been hindered by their inability to purify and chemically characterize their metabolites, despite the fact that many of these microorganisms have been demonstrated to include them in their culture filtrates [4,5].
Plant pathogen metabolism generates non-enzymatic substances known as phytopathogenic toxins, which are toxic to plants [6]. These poisons have the ability to completely kill a plant’s regular physiological processes at very low concentrations. Toxins produced by phytopathogenic fungi can have a significant impact on the emergence of plant diseases and consequently harm the host plants [7]. Most phytopathogenic toxins are secondary metabolites with low molecular weight that can cause wilting, growth suppression, chlorosis, necrosis, and leaf spotting, among other particular symptoms [8]. Toxins that are phytopathogenic have a complicated mode of action. It primarily affects the host plant’s cell membrane, mitochondria, and chloroplasts (Cp), which kills the plant or disrupts with its metabolism [9]. Furthermore, it prevents the host plant from synthesizing proteins and nucleic acids, which can lead to physiological problems, cell death, and ultimately the plant’s own demise [10]. Furthermore, it prevents the host plant from synthesizing proteins and nucleic acids, which can lead to physiological problems, cell death, and ultimately the plant’s own demise [10]. Understanding how pathogens and plant hosts interact, as well as how to use pathogenic toxins to detect plant disease resistance, find disease-resistant mutants, and management of plant diseases, all depend greatly on how well we are able to study the mechanisms of pathogenic toxins in pathogenicity [11-14]. Reviews of phytopathogenic microbes and their toxins have been few thus far [9].
Several plant pathogens produce toxins that harm host plants. Some toxins can cause almost all the peculiar symptoms of a plant disease [15]. These toxins (secondary metabolites) are deadly to plants even at very low concentrations, and many of them intensify fungal or bacterial disease symptoms [16]. Few toxins show increased toxicity only against the hosts of toxin-producing pathogens whereas in some cases, a pathogen’s potential to produce toxin is related to its disease-causing ability [17]. Toxins are host specific or non-host specific. Few toxins are active on many plant species and are considered as non-host selective, while some of the toxins act on one species, called host-specific toxins, like victorin. Yoder (1980) classified plant pathogen toxins as a pathogenic or virulence factor based on their involvement in disease development [18].
Toxin has been involved in pathogenesis all the way back to Anton de Bary, who strengthened the theory of plant disease generally called as “theory of toxins.” The theory of toxin suggests that a pathogen-induced toxin causes all disease symptoms. This theory was largely refuted, as more information about plant diseases was gathered. Afterward, the toxin theory of plant diseases was reinvigorated by the discovery of the host-specific toxin like victorin. The toxin can harm or kill the host by directly affecting the living host cells [19].
2. CLASSIFICATION OF TOXINS
According to Wheeler and Luke [20], there are three types of toxins, namely phytotoxins, vivotoxins, and pathotoxins as mentioned in Table 1.
Table 1: Difference between pathotoxin, phytotoxin, and vivotoxin with examples [21].
| Pathotoxins | Phytotoxins | Vivotoxins |
|---|---|---|
| Discovered by Wheeler and Luke, 1963 [20]. | Discovered by Wheeler in 1975 [22]. | Discovered by Dimond and Waggoner, 1953 [23]. |
| Pathotoxins are toxins that play a significant part in causing plant disease. They are secreted by the plant pathogen or by the contact between host and pathogen. | Phytotoxins are the toxic compounds secreted by living organisms and their role in disease is simply suspected rather than confirmed. | Vivotoxins are secreted in the diseased host by either the pathogen or its host plant. |
| Pathotoxins, when utilized to a sensitive host at low concentration, should either show all or most of the symptoms of the plant disease. The host range and resistance/susceptibility spectrum of the toxin and the pathogen ought to be identical. The pathogenicity should be directly related to the pathogen’s ability to secrete toxin. | The function of phytotoxins in plant disease development is suspected because the release of the toxins and the pathogenicity of the disease-causing agent have no relationship. | Vivotoxins have a partial significance in the cause of plant disease. Reproducible isolation of the toxin from the infected host and induction of at least some of the disease symptoms when inoculated to a healthy plant. |
| They may be non-specific or specific. | They are non-specific. | These are generally non-specific. |
| Examples:- Tab-toxin: Pseudomonas tabaci HMT toxin: Drechslera maydis race T | Examples:- Alternaric acid: Alternaria solani Cochliobolin: Cochliobolus spp. | Examples:- Fusaric acid: Fusarium spp. Pyricularin: Pyricularia oryzae |
3. EFFECT OF TOXINS ON PLANT TISSUES
3.1. Changes in Cell Wall Permeability
All diseased plant tissues show changes in cell wall permeability [24]. Some show this effect before any visible symptom of the disease. Some toxins kill plant cells by altering plasma membrane permeability, allowing water and electrolytes to leak out and the toxin to enter [25]. Cell wall permeability is affected by lycomarasmin, fusaric acid, picolinic acid, and victorin [26,27]. In the case of victorin, even at very low concentrations, the toxin makes sensitive tissues more permeable, which causes the cells to leak. As a result, they cannot store salts and other substances. Similar results can be acquired with resistant plants but only when the toxin concentration is high. Gaumann (1958) believed that leaf cell permeability was a primary factor in wilt diseases. In tomato plants, fusaric acid affects the semi-permeability of the plasma membrane, allowing metal ions, amino acids, and peptides to escape in the cell’s transpiration stream and damaging osmotic and turgor pressure [28]. Gottlieb observed the fluid in tomato plants attacked by Fusarium lycopersici enhanced marrow cell water permeability by nearly 3 times higher than the healthy tomatoes [29].
3.2. Disruption of Normal Metabolic Process of Plants
Toxins or antibiotics intervened with enzyme systems and produced derangement of vital metabolites [30]. Linskens detected K, Ca, Na, and amino-acid leakage from lycomarasmin and fusaric acid-affected leaves. Many physiological activities of host cells are disrupted due to changes in cell wall permeability. The disturbed salt balance in the protoplasm causes an increase in respiration [31]. Loss of water and other substances cause enzyme system malfunction, resulting in cell death. Gnanam (1956); Sadasivan and Saraswathi-Devi (1957); and Sadasivan and Kalyanasundaraan (1959) reported ionic imbalance in Fusarium-infected cotton plants [32-34]. Pyricularin inhibits the polyphenol oxidase system, and victorin decouples oxidative phosphorylation. With heavy metal ions, fusaric acid and picolinic acid can create chelating complexes, inhibiting the enzyme system that needs those metals to function. Toxin-resistant tissues either inactivate the toxin or have an alternative enzyme system for normal function [35].
Obstruction with the growth regulator system of the plant may cause stimulation of the growth of plant parts. Conversely, few toxins interfere root growth such as Fusarium moniliforme that gives rise to a thermostable toxin even in the soil around the roots, which causes roots’ browning and restricted development. The toxin also produces plant growth regulating and phytotoxic impact on plant system [36].
4. TOXINS SELECTIVELY SPECIFIC TO HOST
4.1. Victorin (HV)-Toxin
Victorin was one of the first HSTs discovered, and its toxicity (effects at 10 pM), rapid cell effects, and high specificity made it the archetype HST [37]. As a consequence of the development and extensive usage of the Victoria oat variety and its derivatives, the fungus Cochliobolus (Helminthosporium victoriae) was 1st time visible in 1945–1946 and has been the cause of it [38]. The Vb gene for resistance to crown rust, which is infected by Puccinia coronata f.sp. avenae, was present in these oats. The Vb gene has not been genetically distinguished from Pc-2, a gene for resistance against crown rust pathogen [39,40], indicating that the two genes may be identical.
Victorin’s ability to operate as a particular elicitor for the generation of phytoalexins, known as avenanthramides, in oat cultivars expressing the Pc-2 gene is what makes it so intriguing [41,42]. Cochliobolus victoriae infects sensitive oat plants’ basal portion, produces toxin then inoculated to the leaves, and resulted in plant death, whereas other varieties of oat’s and other plants that were evaluated were either resistant to the fungus and toxin or were moderately sensitive to it. Producing toxins in mushrooms is controlled by a single TOX3 gene. The toxin causes numerous changes at histological and metabolic levels in the host, such as structural alterations of the cell wall, cellular electrolyte losses, enhanced respiration, reduced development, and protein synthesis, in addition to other visible symptoms of disease brought on by the pathogen. Ion, salt, and acid leakage from the tonoplast of mitochondria cause increased respiration in victorin-treated tissues [43].
Furthermore, in culture, only toxin-producing isolates of fungus are pathogenic to oats, while non-pathogenic isolates do not secrete toxins. Victorin is a complex, partially cyclic chlorinated pentapeptide [38]. Victorin is highly mobile in plants [44]. Victorin appears to bind to various proteins in the cell plasma membrane, but how it affects susceptible plants’ metabolism remains unknown [45]. Victorin primarily affects cell wall permeability [20].
Victorin inhibits the photorespiratory enzyme glycine decarboxylase, which cleaves RUBISCO and leads to cell death by exchanging protons between Cps, mitochondria, and peroxisomes as mentioned in Figure 1 [46]. Victorin also causes laddering of DNA and condensation of heterochromatin, the common signs of apoptosis [47-49]. Victorin shrinks cells and lowers mitochondrial membrane potential, according to Curtis and Wolpert (2004), without compromising the integrity of the plasma membrane or the tonoplast [50]. Animals undergo Victorine-like apoptosis as a result of calcium ion efflux, oxidative burst, serine and cysteine proteases, and nuclease activity [47-49]. Therefore, transcriptional and/or metabolic reprogramming of host cells, which results in the stimulation of a cellular suicide pathway, is probably what causes victorin-induced cell death [45].
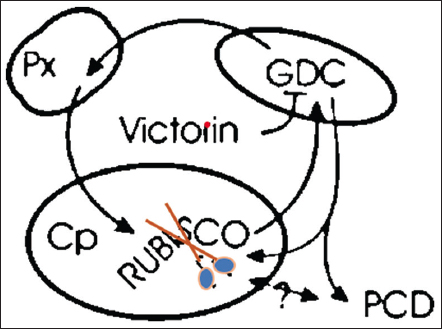 | Figure 1: Victorin is an exceptionally potent inhibitor of glycine decarboxylase, which is crucial to the photorespiratory cycle. This cycle involves the interchange of products from the RUBISCO oxygenase reaction between the peroxisome, mitochondrion, and chloroplast. Treatment with victorin causes RUBISCO to cleave and causes programmed cell death (PCD). How the RUBISCO cleavage connects to the PCD mechanism is unknown. [Click here to view] |
4.2. T-Toxin or Cochliobolus heterostrophus Toxin (HMT Toxin)
The C. heterostrophus (T-variety) produced the T-toxin and is a contributing factor to the exceptional virulence of Texas male sterile cytoplasm-bearing maize and is the cause behind Southern Corn Leaf Blight (SCLB) [51]. There are two pathogenic varieties of C. heterostrophus. In 1925, Race O was previously identified as a maize pathogen [52]. Race T was first developed in the US in 1968. It is identical to other C. heterostrophus breeds other than its capacity to manufacture T toxin. By 1970, it only targeted Texas male corn in the Corn Belt [53]. Three decades later, we are aware that the Toxl locus, which is absent from the O race of C. heterostrophus, is the primary distinction between the T and O races of this organism. Since the T race is derived from race O [54-56], a molecular knowledge of the Toxl locus in T and O must advance the knowledge of how a new pathogenic fungus race evolves [57] whereas, corn with normal cytoplasm is fungus as well as toxin resistant. Both C. heterostrophus T susceptibility and resistance are received from the mother (in the cytoplasmic genes). The potential of C. heterostrophus T to secrete T-toxin and its pathogenicity in TMS maize are both controlled by an identical gene. T-toxin improves virulence but is not essential for the disease causing ability of C. heterostrophus race T [51].
T-toxin is a blend of long linear polyketols (35–45 carbon) with a 41-carbon main component [58]. The toxin acts on the mitochondria of sensitive cells, rendering them defective and inhibiting synthesis of ATP. T toxin binds to a protein molecule (URF 13) on sensitive inner membrane of mitochondria as shown in Figure 2 [59,60]. Plants with Texas-type cytoplasmic male infertility may contain a minute rearrangement in their mitochondrial chromosome encoding the URF13 protein (T-URF13 gene). Normal maize cytoplasm lacks this gene and protein. Yoder and Gracen (1975) found that in maize lines infected with cytoplasmic male sterility, the presence of T toxin causes the URF13 protein to generate pores in the inner mitochondrial membrane [61], which results in diffuse tissue necrosis, presumably by apoptosis [62]. The pores create the loss of integrity of mitochondria, mitochondrial membrane, selective permeability, and cause disease [63].
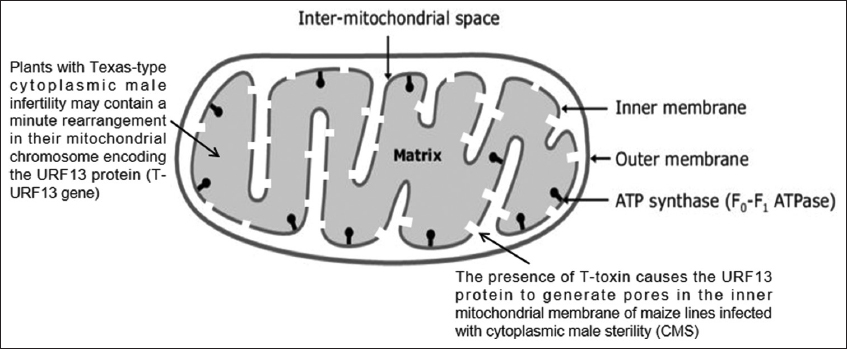 | Figure 2: T-toxin binds to a protein molecule (URF 13) on sensitive inner membrane of mitochondria. [Click here to view] |
4.3. HC-Toxin or Helminthosporium carbonum Toxin
Cochliobolus carbonum causes leaf spot of maize and secretes host-specific toxin HC, which shows toxicity only on selective maize lines, was isolated and crystallized by Pringle and Scheffer [64]. Although the HC toxin’s exact mode of action is unknown, however, it is the only toxin whose genetic, pharmacological, and molecular origins are known. The HC toxin has a molecular formula that approaches C32H50N6O [65]. A gene (Hm1) in resistant maize lines produces the HC-toxin reductase enzyme, which lowers and causes detoxification of the toxin [Figure 3]. Sensitive maize lines are tolerant to the HC toxin despite lacking Hm1 gene. According to experimental findings, HC toxin exerts its effects by delaying the onset of gene expression alterations required to establish responses to artificially induced differences. The secondary metabolic abnormalities seen with H. victoriae toxin are not present in corn tissue inoculated with H. carbonum toxin [66]. It loses its unique toxicity due to its relative instability. Reduced water solubility and nitrogen loss seem to be connected to this activity loss [65].
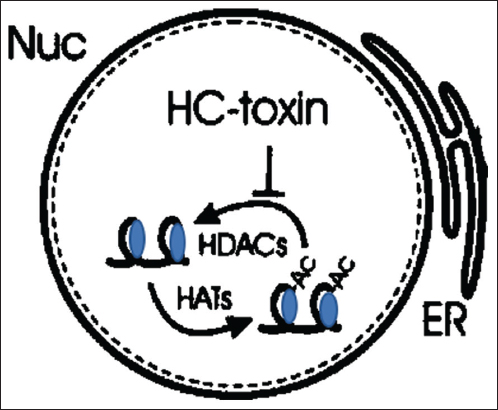 | Figure 3: Histone deacetylases are known to be inhibited by HC-toxin in the nucleus (Nuc), which leads to a hyperacetylation of histones through histone acyltransferases. It is believed that this causes alterations in gene expression that stops antifungal chemicals from being synthesized. [Click here to view] |
4.4. HS-Toxin or Helminthosporoside
The toxin’s structure is reported as a 2-hydroxycyclopropyl-a-D-galactopyranoside [67]. Helminthosporium sacchari, causing eye spot disease, is particularly virulent on seedlings of specific sugarcane cultivars [68]. The host-selective toxin secreted by the disease-causing fungus causes reddish-brown streaks on sensitive leaves [69]. The toxin alters Cps and cell wall permeability in sensitive hosts but not in resistant ones. The protein in resistant lines has no affinity for the toxin [68].
They did not, however, conduct synthesis to verify this structure. The history of leaf temperature seems to have an impact on toxin sensitivity. The toxin is made tolerable by brief foliar pre-treatments at temperatures above or below the growth temperature. This study also shows that toxin generation in pure fungal crops cultivated in the existence of serinol (2-amino-1, 3-propandiol) is expanded significantly to 1,000 times when the fungus is cultivated amid sugarcane suspension crops from disease-susceptible or resistant varieties [70].
4.5. PC-Toxin or Periconia Toxin
Milo disease or Periconia blight caused by a soil-borne pathogen Periconia circinata attacks only milo type of grain sorghum (Sorghum vulgare var. subglabrescens). In 1948, Leukel established that the milo disease was caused by a toxin that was host specific. Only virulent strains produce a low-molecular-weight polypeptide toxin [71]. Two host-selective polypeptide toxins, called A and B, were extracted from the culture filtrates [64].
Toxin A, the main toxin (molecular weight <2000), has a non-amino acid fraction. It induces metabolic change in sensitive corn tissue in a similar way to that of the H. victoriae toxin [72]. There is evidence that it contains three or more related and selectively toxic compounds with different chemical properties. Acid hydrolysis of one of these compounds produced alanine, aspartic acid, glutamic acid, and serine in a ratio of 6:4:2:2. In structure and biological activity, it appears equivalent to T-toxin. The toxin causes rapid loss of K+-ions and other materials through leakage of the plasma membrane of susceptible tissues but has no effect on resistant tissues. It does not act on isolated mitochondria, Cps, and nucleic activities and directly affects protein synthesis [73].
4.6. AM-Toxin or Alternaria mali Toxins
The Alternaria alternata pathotype that causes the Alternaria apple leaf spot (formerly known as A. mali) produces AM-Toxins [74,75]. The newly discovered and described AMT gene, which is only found in the A. alternata apple pathotype isolates, plays a critical part in the production of the AM toxin. Two AM toxin biosynthetic genes are designated, namely AMT1 and AMT2 [74,76]. Non-ribosomal peptide synthetase is encoded by AMT1, and aldo-keto reductase is encoded by AMT2 [76,77]. The crucial cell organelles known as Cps are where AM poisons mostly cause tissue damage [Table 2][78].
Table 2: List of A. alternata-produced host-specific toxins known to date.
| Alternaria pathotype | Toxin Produced | Target site of toxin | Type of disease and host | Genes Involved | Chemical Structure/Characteristics | Reference |
|---|---|---|---|---|---|---|
| Apple pathotype (Alternaria mali) | AM-toxin Type-I Type-II Type-III | Membrane protein; chloroplasts | apple blotch Apple | AMT genes | Cyclic peptide | [95,96] |
| Sunflower pathotype (A. alternata) | AS-toxin Type-I | Not known | Leaf spotting Sunflower | Not known | Tetrapeptide | [97] |
| Pathotype of Japanese pear (Alternaria kikuchiana) | AK-toxin Type-I Type-II | Sulfhydryl-bearing molecules in membrane protein | Black spotting Japanese pear | AKT genes | Epoxy-decatrienoic esters | [98-100] |
| Rough Lemon pathotype (Alternaria citri jambhiri) | AC-toxin ACR-toxin Type-I | Mitochondria | Leaf spotting citrus rootstocks | ACRT genes | Terpenoid | [101,102] |
| Tangerine pathotype (Alternaria citri tangerine) | ACT-toxin Type-I Type-II | Membrane proteins | Brown spotting Tangerines and Mandarins | ACTT genes | Epoxy-decatrienoic esters | [100,103] |
| Strawberry pathotype (Alternaria alternata f. sp. fragariae) | AF-toxin Type-I Type-II Type-III | Microsomal phospholipase A2 | Black spotting Strawberry | AFT genes | Epoxy-decatrienoic esters | [104,105] |
| Tobacco pathotype (Alternaria alternata f. sp. longiceps) | AT-toxin Type-I Type-II | Mitochondria | Brown spotting Tobacco | ATT genes | Not known | [95,106] |
| Alternaria jesenskae and Cochliobolus (Helminthosporium carbonum) | HC-toxin | Mitochondrial membrane | Leaf spotting and ear rotting Zea mays | A. jesenskae TOXE, A. jesenskae TOXA, A. jesenskae TOX2 genes | Cyclic tetrapeptide | [107,108] |
| Tomato pathotype (A. alternata f. sp. lycopersici) | AL-toxin Type-Ta Type-Tb | Aspartate carbamoyl transferase; sphinganine N-acltransferase | Stem canker Tomato | ALT genes | Aminopentol esters | [109-112] |
| Brassica pathotype (A. brassicicola) | AB-toxin | Not known | Black leaf spotting Brassica species | Not known | Protein | [113,114] |
| A. brassicae | Destruxin-A, Destruxin-B, ABR-toxin | Vacular H+-ATPase | Gray leaf spotting Brassica species | DtxS genes | Not known | [115-118] |
| Peach pathotype (A. alternata) | AP-toxin | Not known | Black spotting Peach | Not known | Not known | [119] |
| A. panax | AP-toxin | Not known | Alternaria blight American ginseng | Not known | Not known | [120,121] |
| Knapweed (A. alternata) | Maculosin toxin | Ribulose-1, 5-bisphosphate carboxylase | Black leaf blight knapweed | Not known | Cyclic peptide | [122,123] |
A. alternate: Alternaria alternata
The cyclic depsipeptide toxin molecule often exists as a combination of three forms [79,80]. Other plant pathogen toxins, such as the HC toxin of C. carbonum race1 [81] and the tentoxin of Alternaria tenuis, also contain this type of structural chemical compound of the cyclic depsipeptide [82]. The resistant apple cultivars are able to withstand more than 10,000 times the normal amount of toxins without developing any symptoms. Sensitive cells’ plasma membranes grow in the infoldings as a result of the AM toxin, and the cells exhibit substantial electrolyte loss. The interface between the cell wall and the plasma membrane is where toxins first emerge to be toxic, but the AM toxin also causes a rapid loss of chlorophyll [Figure 4], indicating that this toxin has multiple sites of action [83].
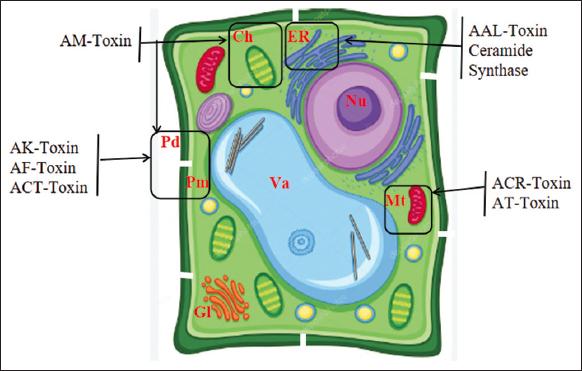 | Figure 4: Schematic presentation of target sites of host-specific toxins (HSTs) produced by Alternaria alternata. Ch, chloroplast; ER, endoplasmic reticulum; Gl, Golgi apparatus; Mt, mitochondrion; Nu, nucleus; Pd, plasmodesmata; Pm, plasma membrane; Va, vacuole. [Click here to view] |
4.7. AK-Toxin or Phyto Alternarin
Alternaria kikuchiana produced the AK-toxin that causes the black spot of the Japanese pear (Pyrus serotina). The pathogen only affects the Nijissciki cultivar and its descendants [Table 2]. Tanaka [84] made the very first mention of toxin’s role in disease causing and latterly by other workers in Japan [85-89]. Three selective toxins designated as phyto-alternarin A, B, and C have been obtained from the culture filtrates of the fungus [90].
The mechanism of action of AK-Toxin is similar to that of victorin. Scientists isolated an HST called AK-toxin, simultaneously monitoring host selectivity during isolation, and found that their toxin preparation satisfied the HST criteria [91-94]. The same preparations of AK toxin were always extracted from filtrates of pathogenic isolate cultures from different regions of Japan. The AK toxin preparation was highly toxic to ten cultivars, including Nijisseiki, but not to 26 other cultivars of pears and non-host plants. This spectrum of toxicity in Japanese pear cultivars coincided with pathogen susceptibility. All A. alternata isolates that produce AK toxin may cause black spots on sensitive pear leaves [93,94]. AK toxin was released by germinating pathogen spores, but no free AK toxin was detected in the dormant spores. Toxin AK caused venous necrosis on leaves sensitive to 0.01 μg/mL but not on leaves resistant even to 100 μg/mL [Figure 4].
4.8. PM-Toxin
In conjunction with SCLB, a new disease, Yellow Leaf Blight of Maize [124], caused by Mycosphaerella zeae-maydis [125], has been recognized. The SCLB outbreak of 1970 devastated maize fields due to T-race of C. heterostrophus. A second fungus, Phyllosticta zeae-maydis, with the same biological specificity, appeared by coincidence. Race T produces toxin T, while Phyllosticta zeae-maydis produces PM-toxin [109,126], both host-selective polyketide toxins required for super-virulence [127]. M. zeae-maydis is also specific for maize T and produces the toxin PM [128,129]. Host-specific maize pathotoxins with linear C33 or C35 poly-ketol structures were generated by Phyllosticta maydis [130].
5. NON-SPECIFIC TOXINS
5.1. Tabtoxin
Tabtoxin has been first reported by Johnson and Murwin [131] and confirmed by Clayton [132] to be produced by the bacterium Pseudomonas syringae pv. tabaci, which causes the wildfire disease of tobacco. Pathovar tabaci strains can be found on different hosts, like beans and soybeans [133,134], and by other pathovars of P. syringae, that are found on coffee, maize, and oats. Necrotic spots are caused by toxin-producing strains on leaves, and each spot has a yellow halo encircling it. Not only do sterile culture filtrates of the organism and pure toxin elicit symptoms similar to tobacco wildefire, but they also do so in a wide range of plant species from other families [135]. Oftentimes P. syringae pv. tabaci strains yield mutants (Tox-) that are incapable of producing the toxin. Tox-strains create necrotic leaf spots lacking a yellow halo and have decreased virulence. Pseudomonas angulata, the causative agent of tobacco angular leaf spot, is indistinguishable from toxic strains and is currently believed to be a non-toxic variant of P. syringae pv. tabaci [136].
Wooley et al. [137] reported the first isolation of tabtoxin, and Stewart [138] published an appropriate structure. The common amino acid threonine and the hitherto unidentified amino acid tabtoxinine combine to form the dipeptide known as tabtoxin. The active toxin, tabtoxinine, is released when the inactive toxin, tabtoxin, breaks down within the cell. The structure of tabtoxin is composed of tabtoxinine-b-lactam [2-amino-4-(3-hydroxy-2-oxoazacyclobutan-3-yl)butanoic acid] and threonine. A different kind of tabtoxin known as [2-serine] tabtoxin [139] is also created in trace amounts as an analog that has a serine molecule substituted in place of a threonine molecule. Both forms of tabtoxin are produced in a physiologically inert state, but they can be easily transformed into the active tabtoxinine-b-lactam moiety by cleaving serine or threonine using certain aminopeptidases found in the plant or bacteria. The target enzyme glutamine synthetase, which catalyzes the conversion of glutamic acid to glutamine in amino acid metabolism, is inhibited by the active moiety tabtoxinine-b-lactam [Figure 5]. The aforementioned inhibition causes an aberrant build-up of ammonia in tobacco cells, which gives rise to the recognizable chlorosis [140].
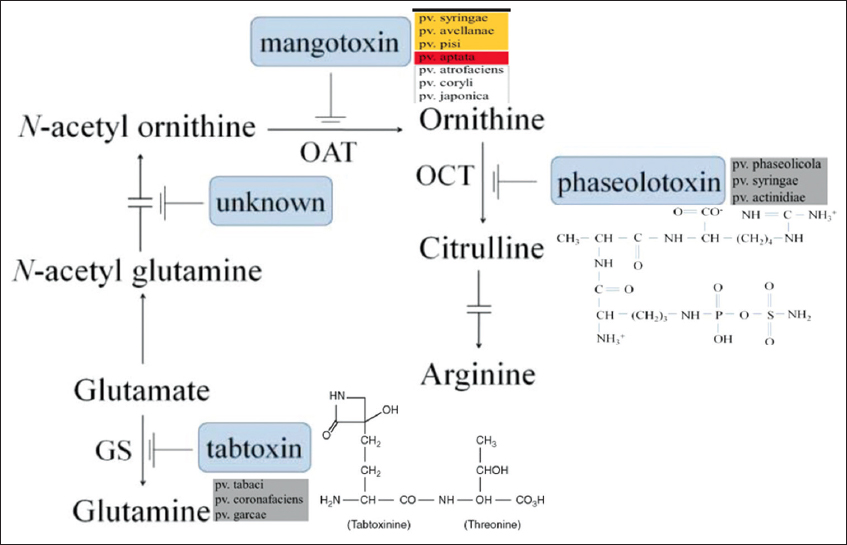 | Figure 5: The production of mangotoxin and its enzymatic target. An illustration of the processes involved in the manufacture of polyamines and arginine-glutamine. The several pathovars responsible for the synthesis of the various antimetabolite toxins are demonstrated along with the enzymatic targets of the various antimetabolite toxins, including mangotoxin. OAT, ornithine N-acetyltransferase; OCT, ornithine carbamoyltransferase; ODC, ornithine decarboxylase; and GS, glutamine synthetase [Click here to view] |
5.2. Phaseolotoxin
Phaseolotoxin (N-sulfodiaminophosphinyl-ornithyl-alanyl-homoarginine) is a phytotoxic secondary metabolite produced by P. syringae pv. phaseolicola that causes halo blight of beans [141]. The primary hosts are lima beans, red kidney beans, cranberry yellow beans, green beans, scarlet runner, kudzu vine, and Proteus vulgaris common [142]. Sawada and Fujikawa reported that this toxin is also produced by P. syringae pv. actinidiae, the causative agent of kiwifruit bacterial cancer [143].
Infected plants exhibit both localized and systemic chlorotic symptoms at the same rates. These symptoms were resulted in restricted growth of freshly expanded leaves, regulated apical dominance, and ornithine production. Systemic infections are uncommon but more frequently manifest in some dry bean varieties as juvenile leaflets that curl, become yellow, and eventually die [144]. The Pht region, an island of pathogenicity, contains 23 genes arranged in five transcriptional units: Two monogenic units (argK, phtL) and three arranged as operons (phtA, phtD, phtM), the majority of which have unidentified functions. The products of a few of these genes are involved in the production of phaseolotoxin [141].
By interacting with the active site and deactivating the ornithine carbamoyl transferase enzyme, which ordinarily transforms ornithine into citrulline, a precursor of arginine, the toxin damages cells as shown in Figure 5 [145]. Phaseolotoxin, however, also appears to inhibit the biosynthesis of pyrimidine nucleotides, reduce ribosome activity, interfere with lipid synthesis, modify membrane permeability, and cause the accumulation of large starch granules in Cps [83].
5.3. Tentoxin
The A. alternata (formerly known as A. tenuis), which was first described by George Templeton et al. [146], is the source of tentoxin. It is a cyclic tetrapeptide that is documented for binding with Cp-coupling factor, and involved in the process of energy transfer in Cps by activating it. Additionally, it inhibits the conversion of ADP into ATP that occurs under the influence of light [147]. Tentoxin interferes with the normal growth of Cps and causes chlorosis in species that are sensitive to its effects. It leads to the production of less chlorophyll. Additionally, but unrelatedly, tentoxin suppresses the action of polyphenol oxidases, which are involved in a number of plants defense systems [148].
5.4. Cercosporin
Cercospora and numerous other fungi produced cercosporin toxin. It is associated with the infections and leaf spots that appear on a wide variety of cultivated plants, such as Cercospora zinnia leaf spots and gray corn leaf spots [149]. A photosensitizing perylenequinone called cercosporin absorbs light energy, transforms it into an energetically active state, and combines with molecular oxygen to create activated oxygen, which damages host plant membranes and feeds the intercellular pathogen by providing nutrients. The toxicity of photosensitizers is due to oxidative damage to a plant cell’s lipids, proteins, and nucleic acid and kills plants, thus increasing the pathogen’s virulence [150]. Fungal spores and mycelium produce pyridoxine (vitamin-B6), which neutralizes it by combining with one oxygen atom and allowing them to withstand against the cercosporin toxicity [151].
Cercosporin production is mediated by multiple signal transduction pathways [152]. Calcium and calmodulin signaling may be involved in the generation of cercosporin, according to studies utilizing pharmacological inhibitors [153]. The development of cercosporin in C. zeae-maydis was also linked to a gene encoding a MAP kinase, according to a study [154]. Additionally, the transcription factor CRG1 controls the production of cercosporin [155].
5.5. Tagetitoxin
P. syringae pv. tagetis, which produces this toxin, was first identified as a pathogen affecting calendula production in Denmark in 1955 [156]. It is also the cause of bacterial leaf spots and apical chlorosis in several species of the Compositae family, including African marigold (Tagetes erecta L.), sunflower (Helianthus annuus L.), common ragweed (Ambrosia artemisiifolia L.), Jerusalem artichoke (Helianthus tuberosus L.), dandelion (Taraxacum officinale Weber), bush plant (Silphium perfoliatum L.), and another species of sunflower (Helianthus salicifolius A. Diter) [157-161]. Mitchell and Durbin [162] first postulated an eight-membered ring as the chemical structure of the TGT; however, this was soon changed to a two cyclic structure on the basis of NMR and mass spectrometry (shown right) [163]. Tagetitoxin has the chemical formula - C11H17N2O11PS [162].
The toxin results in unexpected apical chlorosis, necrotic leaf patches, and even a chlorotic halo. It functions by preventing Cp RNA polymerase III from acting [164,165]. Based on their capacity to create tagetitoxin, pathogens are categorized into three classes: Class 1 and 2 strains may cause the toxin to be produced in plants, but Class 3 strains cannot [166].
5.6. Fusicoccin (FC)
Fusicoccum amygdali causes almond and peach twig rust that causes the leaves of an infected shoot to wilt and dries out and causes cancer around infected buds and nodes [167]. The phytotoxin called FC produced by fungi induces symptoms of downy mildew when introduced into the xylem [168]. The toxin is a carbotricyclic terpene glycoside [169]. The main metabolite FC A induces the wilt in the plant tissues at concentrations up to 0–1 μg/mL, currently undergoing phytotoxicity tests. FC toxin B, C, and D and aglycone are comparatively very less phytotoxic than FC A. The phytotoxicity of FC A is non-selective, although highly active when given through the vascular system [170].
FC affects cellular transport systems and increases the absorption of several anions, sugars, and amino acids by affected cells. The toxin is regarded as the first clear example of wilt toxin because it has been shown to reproduce physiological causes of wilt, not just the wilt symptoms. It also elevates opening of stomatal, respiration, and enlargement of cell [171].
5.7. Marticin
It is a pathotoxin produced by Fusarium sp. of the Mortierella group. Highly pathogenic strains of the pea pathogen, Fusarium solani f.sp. pisi, produce large quantities of a marticin [172]. In detail, marticins toxin was initially inspected by Hardegger and Pfiffner. The two compounds, marticin, and isomarticin are acquired from the acid part of extract of fungus in which the isomarticin predominates [173].
Less pathogenic strains produce less phytotoxic compounds javanicin and fusarubin. However, marticin was obtained from diseased plant tissues in sufficient amount to induce wilted necrosis [174]. Kern proposed that the phytotoxic action of marticin could be due to its inhibition of malate or citric acid dehydration in the citric acid cycle [175].
5.8. Lycomarasmin
Tomato wilt is solely caused by Fusarium oxysporum f.sp. lycopersici. Lycomarasmin and fusaric acid were discovered filtrates of the culture of fungus causing tomato wilt by Gaumann et al. in 1952 [176]. Tomato wilt symptoms mimic the toxic damage caused by the combined effects of these two toxins. The researchers also mentioned the wilt toxin vasofuscarin, which is thought to contribute to the progression of disease [177]. Lycomarasmin is a polypeptide having the empirical formula C9H15O7N3 and a molecular weight of 277.3. Lycomarasmin was proposed by Woolley (1948) as the modified tripeptide [178].
Waggoner and Dimond (1953) reported that the toxin inactivates the growth factor strepogenin, which counteracts its effects. After being applied to tomato cuttings, purified lycomarasmin works as a chelating agent and creates an iron-containing, water-soluble, unstable complex that is transported to the leaves [179]. Common symptoms are brought on by iron that is released into the leaves. Iron makes the poison more toxic, whereas copper makes it less hazardous. Enhanced water permeability of the epidermis and increased transpiration of tomato cuttings are brought on by lycomarasmin in the presence of iron [180].
5.9. Fusaric Acid
The existence of this toxin was originally noted in 1934 from Fusarium heterosporum; nearly 20 years later, Gauman et al. (1952) also observed its presence from F. oxysporum f.sp. lycopersici, F. oxysporum f.sp. vasinfectum, and Gibberella fujikuroi, recognized its poisonous character [176]. After that, numerous Fusarium formae specialis of the elegance group, including F. oxysporum f.sp. lycopersici, batatis, conglutinans, cubense, lini, vasinfectum, udum, and F. moniliforme, have been found to contain this phytotoxic metabolite [181-185].
It is categorized as a non-specific vivotoxin and is the most researched wilt toxin produced by a pathogen. It does not, however, manifest every indication of wilt. It is now widely acknowledged that fusaric acid is not a byproduct of autolysis but rather is created during the intense development phase [186]. Fusaric acid is secreted by developing hyphae, although the majority of it is released after mycelial autolysis begins [187,188].
Additionally, in the tissue extract, direct screening of this metabolite of afflicted plants has been performed and has been successful to a significant degree [189]. It has also been noted that some species produce the toxin in the rhizosphere soil of tomato plants [190]. Contrarily, some publications [183] claim that although the same pathogen secretes fusaric acid in culture solution, not in the host tissues. They added that this toxin did not contribute to pathogenicity. Fusaric acid is claimed to play a variety of roles in plants. It disturbs the cell wall permeability and leads to the complex formation of copper and iron in the host cell tissues. This throws off the cell’s ionic balance and enzymatic activities. By enzymes chelation or making the respiration-related enzymes inactive, it changes the plant’s breathing pattern [191].
5.10. Pyricularin
Blast rice disease, caused by Pyricularia oryzae (Magnaporthe grisea), is characterized by leaf spots, collar, and culm rot [192]. Rice crop infected with P. oryzae can release up to nine toxins, depending on the fungal strain and the host range. Pyricularin is more toxic to the fungus itself than to the host plants, and the fungus produces a specific protein to inactivate the antifungal property of the toxin, but not the phytotoxic property [193-195]. Since picolinic acid affects vulnerable types more than resistant ones, the other toxin, pyricularin, is more active and partially specific. Together with picolinic acid, pyricularin serves as an important factor in blast disease by inhibiting the resistance, or the hypersensitive reaction [196]. Polyphenols and oxidases are increased by the toxin. One of the main rice plant polyphenols, chlorogenic acid, reduces the toxicity [197]. Some other microbial toxins, their influence on the host or tissues, and their mode of action are mentioned in Table 3.
Table 3: List of other microbial toxins, their influence on the host or tissues, and their mode of action [44,198-200].
| Toxin | Pathogen | Host | Chemical Structure/Characteristics | Specificity | Mode of Action | Reference |
|---|---|---|---|---|---|---|
| Alternaric acid, altersolanol, macrosporin, and zinniol | Alternaria solani | Potato and Tomato | Di-basic acid Alternaric acid | Non-specific Phytotoxin | The role is doubtful in disease development, wilting, necrosis, and chlorosis occur | [83,198,199] |
| Colletotin | Colletotrichum fuscum | Anthracnose of Digitalis | Same as alternaric acid, polysaccharide and peptide attributes | Non-specific Phytotoxin | Disrupts cell permeability or affects pectic enzymes | [200,201] |
| Diaporthin | Endothia parasitica | Chestnut | Structure similar to isocoumarins | Phytotoxin | Necrosis of the conducting vessels | [202] |
| Helminthosporal | Helminthosporium sativum | Wheat and Barley | Sesquiterpenoid | Non-specific phytotoxin | Interferes with the respiration of root tissues of wheat and barley | [203,204] |
| Ophiobolin A (formerly known as Cochliobolin) | Helminthosporium oryzae | Rice | Oxalic acid | Non-selective Phytotoxin | Phenol metabolism gets hampered | [83,205] |
| Oxalic acid | Aspergillus niger, Sclerotinia sclerotiorum | Crown rot of Groundnut seedlings | Oxalic acid | Phytotoxin | Necrosis | [198,200,206] |
| Fumaric acid | Rhizopus sp. | Hull rot of almonds causes rotting of the fruit mesocarp | Either fumaric acid or its derivatives | Non-specific Phytotoxin | The toxin extends from the damaged fruits and causes lightening in neighboring leaves and twigs | [207,208] |
| Hydrogen cyanide (HCN) | Unidentified Psychrophilic Basidiomycetous Fungus | White crown rot or snow mold of Alfalfa | HCN | Phytotoxin | Necrosis | [209] |
| Thaxtomins (Thaxtomin A and B) | Streptomyces spp. | Scab of Potato | Amino-acid derivatives | Non-specific Phytotoxin | Plant cell hypertrophy and cell death at levels resembling those in scab lesions on field-infected potato tubers | [210,211] |
| Albicidin | Xanthomonas albilineans | Leaf scald of Sugarcane | Peptide antibiotic | Non-specific Phytotoxin | Interfere with the cleavage -relegation step of ATP-dependent DNA at the gyrase A subunit | [212,213] |
| Syringomycin and Syringostatin | Pseudomonas syringae pv. syringae | Leaf spots and cankers of stone fruits | Cyclic lipodepsinonapeptides | Non-specific | Lysis of cellular membrane resulting in necrosis. | [44,214-216] |
| Coronatine | Pseudomonas syringae pv. atropurpurea, tomato, glycinea, morsprunorum, maculicola | Tomato, canker of cherry and plum, bacterial blight of soybean | a-Amino acid, coronamic acid and coronafacic acid | Non-specific | Light-dependency chlorosis, necrosis and stunting of plant tissues. Hypertrophy of potato tuber tissues and causes loosening of cell wall | [44,217,218] |
| Pseudomonas (Ralstonia) solanacearum toxin | Pseudomonas (Ralstonia) solanacearum | Potato | Polysaccharide | Pathotoxin | Block water flow in the vascular system | [198,200,219] |
| Amylovorin | Erwinia amylovora | Apple and Pear (Fire blight) | 98% Galactose in Polymeric forms and 0.3% protein | Specific Phytotoxin | Causes blighting apple and pear | [200,220] |
| SV-toxins I and II | Stemphylium vesicarium | Brown spot disease of European Pear | Structure of these toxins is still unknown | Specific | Cause veinal necrosis in susceptible cultivars of the pear at low concentration | [221,222] |
| Phomalide, Phomalairdenone and Depsilairdin | Phoma lingam | Lesions on Canola (Brassica napus) | Cyclic depsipeptide | Specific | Causes lesion and blackening of lower stems and decay | [223,224] |
6. CONCLUSION
The toxins described previously play a significant portion in the disease development and able to cause all or majority of the symptoms typical of the particular disease in plants that are susceptible. During pathogenesis, microorganisms produce the majority of these toxins. Hence, it may be concluded that the toxin concept can be applied in controlling the diseases of plants. Once identify the toxic compounds, their structure, nature, and mode of action, it easily counteracts that the effect of the toxin can be done. The toxins can also be used for screening resistant varieties on a large scale in less time. The study of toxins helps to know the cause of pathogenesis at the host’s molecular level. This information will help us in our breeding program of evolving disease-resistant varieties of crop plants. Future research on these toxins will help us better understand and evaluate how plants and pathogens interact, as well as provide us with fresh approaches to disease prevention. Laboratory research on the use of plant-pathogenic microbial toxins is still ongoing. It is anticipated that the application of bioengineering techniques, such as tissue culture and cell engineering, will solve this difficulty and hasten the development of useful uses for natural toxins. All these researches in biotechnology and molecular biology field are to bring novel strategies in disease management.
7. AUTHORS’ CONTRIBUTIONS
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agreed to be accountable for all aspects of the work. All the authors are eligible to be an author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
8. FUNDING
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
9. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
10. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
11. DATA AVAILABILITY
Data is available with the authors and shall be provided upon request.Data is available with the authors and shall be provided upon request.
12. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Graniti A, Durbin RD, Ballio A. Phytotoxins and plant pathogenesis. In:Neuhoff V, Friend J, editors. Cell to Cell Signals in Plants and Animals. NATO ASI Series. Vol. 51. Berlin, Heidelberg:Springer;1991. [CrossRef]
2. Gäumann E. Principles of Plant Infection. London:Crosby Lockwood;1950. 543.
3. Gäumann E. Toxins and Plant Diseases. Vol. 13. United States:Endeavor;1954. 198-204.
4. Evidente A. Specialized metabolites produced by phytotopatogen fungi to control weeds and parasite plants. Microorganisms 2023;11:843. [CrossRef]
5. Peng Y, Li SJ, Yan J, Tang Y, Cheng JP, Gao AJ, et al. Research progress on phytopathogenic fungi and their role as biocontrol agents. Front Microbiol 2021;12:670135. [CrossRef]
6. Thynne E, Saur IM, Simbaqueba J, Ogilvie HA, Gonzalez-Cendales Y, Mead O, et al. Fungal phytopathogens encode functional homologues of plant rapid alkalinization factor (RALF) peptides. Mol Plant Pathol 2017;18:811-24. [CrossRef]
7. Soyer JL, Hamiot A, Ollivier B, Balesdent MH, Rouxel T, Fudal I. The APSES transcription factor LmStuA is required for sporulation, pathogenic development and effector gene expression in Leptosphaeria maculans. Mol Plant Pathol 2015;16:1000-5. [CrossRef]
8. Yin Z, Ke X, Kang Z, Huang L. Apple resistance responses against Valsa mali revealed by transcriptomics analyses. Physiol Mol Plant Pathol 2016;93:85-92. [CrossRef]
9. Shang Y, Xiao G, Zheng P, Cen K, Zhan S, Wang C. Divergent and convergent evolution of fungal pathogenicity. Genome Biol Evol 2016;8:1374-87. [CrossRef]
10. Zeilinger S, Gupta VK, Dahms TE, Silva RN, Singh HB, Upadhyay RS, et al. Friends or foes?Emerging insights from fungal interactions with plants. FEMS Microbiol Rev 2016;40:182-207. [CrossRef]
11. Hamel LP, Nicole MC, Duplessis S, Ellis BE. Mitogen-activated protein kinase signaling in plant-interacting fungi:Distinct messages from conserved messengers. Plant Cell 2012;24:1327-51. [CrossRef]
12. Wirthmueller L, Maqbool A, Banfield MJ. On the front line:Structural insights into plant-pathogen interactions. Nat Rev Microbiol 2013;11:761-76. [CrossRef]
13. Bi F, Barad S, Ment D, Luria N, Dubey A, Casado V, et al. Carbon regulation of environmental pH by secreted small molecules that modulate pathogenicity in phytopathogenic fungi. Mol Plant Pathol 2016;17:1178-95. [CrossRef]
14. Adam A, Deimel S, Pardo-Medina J, García-Martínez J, Konte T, Limón MC, et al. Protein activity of the Fusarium fujikuroi Rhodopsins CarO and OpsA and their relation to fungus-plant interaction. Int J Mol Sci 2018;19:215. [CrossRef]
15. Nazarov PA, Baleev DN, Ivanova MI, Sokolova LM, Karakozova MV. Infectious plant diseases:Etiology, current status, problems and prospects in plant protection. Acta Naturae 2020;12:46-59. [CrossRef]
16. Graniti A. Phytotoxins and their involvement in plant diseases. Introduction. Experientia 1991;47:751-5. [CrossRef]
17. Ghazaei C. Advances in the study of bacterial toxins, their roles and mechanisms in pathogenesis. Malays J Med Sci 2022;29:4-17. [CrossRef]
18. Yoder OC. Toxins in pathogenesis. Annu Rev Phytopathol 1980;18:103-29. [CrossRef]
19. Andersen EJ, Ali S, Byamukama E, Yen Y, Nepal MP. Disease resistance mechanisms in plants. Genes (Basel) 2018;9:339. [CrossRef]
20. Wheeler H, Luke HH. Microbial toxins in plant disease. Annu Rev Microbiol 1963;17:233-42. [CrossRef]
21. Srivastava S. Plant Pathology Compare and Contrast. New Delhi, India:Kalyani Publisher;2019. 182.
22. Wheeler H. Plant Pathogenesis. Amsterdam:Elsevier/North-Holland;1975. [CrossRef]
23. Dimond AE, Waggoner PE. The cause of epinastic symptoms in Fusarium wilt of tomatoes. Phytopathology 1953;43:663-9.
24. Houston K, Tucker MR, Chowdhury J, Shirley N, Little A. The plant cell wall:A complex and dynamic structure as revealed by the responses of genes under stress conditions. Front Plant Sci 2016;7:984. [CrossRef]
25. Ottmann C, Luberacki B, Küfner I, Koch W, Brunner F, Weyand M, et al.Acommon toxin fold mediates microbial attack and plant defense. Proc Natl Acad Sci U S A 2009;106:10359-64. [CrossRef]
26. Dube HC. Studies on permeability changes in cotton (Gossypium hirsutum) caused by Verticillium albo-atrum. Proc Indian Acad Sci B 1971;74:185-93. [CrossRef]
27. Harborne JB. Higher plant-lower plant interactions:Phytoalexins and phytotoxins. In:Introduction to Ecological Biochemistry. 4th ed. London:Academic Press Ltd., Harcourt Brace &Co. Publ.;1993. 318. [CrossRef]
28. Gaumann E. The mechanism of fusaric acid injury. Phytopathology 1958;48:670-86.
29. Gottlieb D. The mechanism of wilting caused by Fusarium bulbigenum var. lycopersici. Phytopathology 1944;34:126-35.
30. Sadasivan TS. Symposium on soil microorganisms and plant well-being. Proc Indian Acad Sci 1955;41B:99-101. [CrossRef]
31. Linskens HF. The influence of toxigenic wilt on the leaf excretions of the tomato plant. Phytopathology 1955;23:89-106.
32. Gnanam P. Conductivity studies in cotton plants infected by Fusarium vasinfectum. Proc Indian Acad Sci 1956;44B:125-9. [CrossRef]
33. Sadasivan TS, Saraswathi-Devi L. Vivotoxins and uptake of ions by plants. Curr Sci 1957;26:74-5.
34. Sadasivan TS, Kalyanasundaraan R. Spectrochemical studies on the uptake of ions by plants. 1. The Lundegardh flame technique of ash analysis of toxin/antibiotic invaded cotton plants. Proc Indian Acad Sci 1959;43B:271-75. [CrossRef]
35. Li S, Akrap N, Cerboni S, Porritt MJ, Wimberger S, Lundin A, et al. Universal toxin-based selection for precise genome engineering in human cells. Nat Commun 2021;12:497. [CrossRef]
36. Cole RJ, Kirksey JW, Cutler HG, Doupnik BL, Peckham JC. Toxin from Fusarium moniliforme:Effects on plants and animals. Science 1973;179:1324-6. [CrossRef]
37. Walton JD. Host-selective toxins:Agents of compatibility. Plant Cell 1996;8:1723-33. [CrossRef]
38. Wolpert TJ, Macko V, Acklin W, Jaun B, Seible J, Meili J, et al. Structure of victorin C, the major host-selective toxin from Cochliobolus victoriae. Experientia 1985;41:1524-9. [CrossRef]
39. Meehan F, Murphy HC. A new Helminthosporium blight of oats. Science 1946;104:413-4. [CrossRef]
40. Meehan F, Murphy HC. Differential phytotoxicity of metabolic by-products of Helminthosporium victoriae. Science 1947;106:270-1. [CrossRef]
41. Mayama S, Tatsuki T, Ueno T, Midland SL, Sims JJ, Keen NT. The purification of victorin and its phytoalexin elicitor activity in oat (Avena sativa) leaves. Physiol Mol Plant Pathol 1986;29:1-18. [CrossRef]
42. Mayama S, Bordin AP, Morikawa T, Tanpo H, Kato H. Association of avenalumin accumulation with co-segregation of victorin sensitivity and crown rust resistance in oat lines carrying the Pc-2 gene. Physiol Mol Plant Pathol 1995;46:263-74. [CrossRef]
43. Black HS, Wheeler H. Biochemical effects of victoria on oat tissues and mitochondria. Am J Bot 1966;53:1108-12. [CrossRef]
44. Singh RS. Role of toxins in plant pathogenesis. In:Introduction to Principles of Plant Pathology. 4th ed. New Delhi:Oxford &IBH Publishing Co. Pvt. Ltd.;2009. 151-62.
45. Tada Y, Kusaka K, Betsuyaku S, Shinogi T, Sakamoto M, Ohura Y, et al. Victorin triggers programmed cell death and the defense response via interaction with a cell surface mediator. Plant Cell Physiol 2005;46:1787-98. [CrossRef]
46. Wolpert TJ, Navarre DA, Moore DL, Macko V. Identification of the 100-kD victorin binding protein from oats. Plant Cell 1994;6:1145-55. [CrossRef]
47. Navarre DA, Wolpert TJ. Victorin induction of an apoptotic/senescence -like response in oats. Plant Cell 1999;11:237-49. [CrossRef]
48. Tada Y, Hata S, Takata Y, Nakayashiki H, Tosa Y, Mayama S. Induction and signaling of an apoptotic response typified by DNA laddering in the defense response of oats to infection and elicitors. Mol Plant Microbe Interact 2001;14:477-86. [CrossRef]
49. Yao N, Tada Y, Park P, Nakayashiki H, Tosa Y, Mayama S. Novel evidence for apoptotic cell response and differential signals in chromatin condensation and DNA cleavage in victorin-treated oats. Plant J 2001;28:13-26. [CrossRef]
50. Curtis MJ, Wolpert TJ. The victorin-induced mitochondrial permeability transition precedes cell shrinkage and biochemical markers of cell death, and shrinkage occurs without loss of membrane integrity. Plant J 2004;38:244-59. [CrossRef]
51. Inderbitzin P, Asvarak T, Turgeon BG. Six new genes required for production of T-toxin, a polyketide determinant of high virulence of Cochliobolus heterostrophus to maize. Mol Plant Microbe Interact 2010;23:458-72. [CrossRef]
52. Drechsler C. Leafspot of maize caused by Ophiobolus heterostrophus n. sp., the ascigerous stage of a Helminthosporium exhibiting bipolar germination. J Agric Res 1925;31:701-26.
53. Hooker AL. Cytoplasmic susceptibility in plant disease. Ann Rev Phytopathol 1974;12:167-79. [CrossRef]
54. Leonard KJ. Races of Bipolaris maydisin the Southeastern U. S. from 1974–1976. Plant Dis Rep 1977;61:914-5.
55. Yang G, Rose MS, Turgeon BG, Yoder OC. A polyketide synthase is required for fungal virulence and production of the polyketide T-toxin. Plant Cell 1996;8:2139-50. [CrossRef]
56. Kodama M, Rose MS, Yang G, Yun SH, Yoder OC, Turgeon BG. The translocation-associated tox1 locus of Cochliobolus heterostrophus is two genetic elements on two different chromosomes. Genetics 1999;151:585-96. [CrossRef]
57. Turgeon BG, Lu SW. Evolution of host-specific virulence in Cochliobolus heterostrophus. In:Kronstad JW, editor. Fungal Pathology. Boston:Kluwer Academic Publishers;2000. 93-126. [CrossRef]
58. Kono Y, Takeuchi S, Kawarada A, Daly JM, Knoche HW. Studies on the host-specific pathotoxins produced in minor amounts by Helminthosporium maydis race T. Bioorg Chem 1981;10:206-18. [CrossRef]
59. Braun CJ, Siedow JN, Levings CS 3rd. Fungal toxins bind to the URF13 protein in maize mitochondria and Escherichia coli. Plant Cell 1990;2:153-61. [CrossRef]
60. Rhoads DM, Levings CS 3rd, Siedow JN. URF13, a ligand-gated, pore-forming receptor for T-toxin in the inner membrane of cms-T mitochondria. J Bioenerg Biomembr 1995;27:437-45. [CrossRef]
61. Yoder OC, Gracen VE. Segregation of pathogenicity types and host-specific toxin production in progenies of crosses between races T and 0 of Helminthosporium maydis (Cochliobolus heterostrophus). Phytopathology 1975;65:273-76. [CrossRef]
62. Wolpert TJ, Dunkle LD, Ciuffetti LM. Host-selective toxins and avirulence determinants:What's in a name?Annu Rev Phytopathol 2002;40:251-85. [CrossRef]
63. Møller IM, Rasmusson AG, Van Aken O. Plant mitochondria - past, present and future. Plant J 2021;108:912-59. [CrossRef]
64. Pringle RB, Scheffer RP. Amino acid composition of a crystalline host-specific toxin. Phytopathology 1966;56:11.
65. Pringle RB. Chemical constitution of the host-specific toxin of Helminthosporium carbonum. Plant Physiol 1970;46:45-9. [CrossRef]
66. Xu D, Xue M, Shen Z, Jia X, Hou X, Lai D, et al. Phytotoxic secondary metabolites from fungi. Toxins (Basel) 2021;13:261. [CrossRef]
67. Steiner GW, Strobel GA. Helminthosporoside, a host-specific toxin from Helminthosporium sacchari. J Biol Chem 1971;246:4350-7. [CrossRef]
68. Larkin PJ, Scowcroft WR. Eyespot disease of Sugarcane:Induction of host-specific toxin and its interaction with leaf cells. Plant Physiol 1981;67:408-14. [CrossRef]
69. Steiner GW, Byther RS. Partial characterization and use of a host-specific toxin from Helminthosporium sacchari on sugarcane. Phytopathology 1971;61:691-5. [CrossRef]
70. Pinkerton F, Strobel G. Serinol as an activator of toxin production in attenuated cultures of Helminthosporium sacchari. Proc Natl Acad Sci U S A 1976;73:4007-11. [CrossRef]
71. Leukel RW. Periconia circinata and its relation to Milo disease. J Agric Res 1948;77:201-22.
72. Wolpert TJ, Dunkle LD. Purification and partial characterization of host-specific toxins produced by Periconia circinata. Phytopathology 1980;70:872-6. [CrossRef]
73. Azhari A, Supratman U. The chemistry and pharmacology of fungal genus Periconia:A review. Sci Pharm 2021;89:34. [CrossRef]
74. Johnson RD, Johnson L, Kohmoto K, Otani H, Lane CR, Kodama M. A polymerase chain reaction-based method to specifically detect Alternaria alternata apple pathotype (A. mali), the causal agent of Alternaria blotch of apple. Phytopathology 2000;90:973-6. [CrossRef]
75. Meena M, Gupta SK, Swapnil P, Zehra A, Dubey MK, Upadhyay RS. Alternaria toxins:Potential virulence factors and genes related to pathogenesis. Front Microbiol 2017;8:1451. [CrossRef]
76. Harimoto Y, Hatta R, Kodama M, Yamamoto M, Otani H, Tsuge T. Expression profiles of genes encoded by the supernumerary chromosome controlling AM-toxin biosynthesis and pathogenicity in the apple pathotype of Alternaria alternata. Mol Plant Microbe Interact 2007;20:1463-76. [CrossRef]
77. Harimoto T, Tanaka T, Kodama M, Yamamoto M, Otani H, Tsuge T. Multiple copies of AMT2 are prerequisite for the apple pathotype of Alternaria alternata to produce enough AM-toxin for expressing pathogenicity. J Gen Plant Pathol 2008;74:222-9. [CrossRef]
78. Zhang CX, Tian Y, Cong PH. Proteome analysis of pathogen-responsive proteins from apple leaves induced by the Alternaria blotch Alternaria alternata. PLoS One 2015;10:e0122233. [CrossRef]
79. Ueno T, Nakashima T, Hayashi Y, Fukami H. Structures of AM-toxin I and II host specific phytotoxic metabolites produced by Alternaria mali. Agric Biol Chem 1975;39:1115-22. [CrossRef]
80. Ueno T, Nakashima T, Uemoto M, Fukami H, Lee SN, Izumiya N. Mass spectrometry of Alternaria mali toxins and related cyclodepsipeptides. Biomed Mass Spectrom 1977;4:134-42. [CrossRef]
81. Gross ML, McCrery D, Crow F, Tomer KB, Pope MR, Ciuffetti LM, et al. The structure of the toxin from Helminthosporium carbonum. Tetrahedron Lett 1982;23:5381-4. [CrossRef]
82. Mayer AM, Staples RC, Gilad NL. Mechanisms of survival of necrotrophic fungal plant pathogens in hosts expressing the hyper-sensitive response. Phytochemistry 2001;58:33-41. [CrossRef]
83. Agrios GN. Plant Pathology. 5th ed. Amsterdam:Elsevier Academic Press;2005.
84. Tanaka S. Studies on black spot disease of the Japanese pear (Pyrus serotina Rehd.). In:Molecular Strategies of Pathogens and Host Plants. Vol. 28. Berlin:Springer;1933. 1-31.
85. Torikata H. Studies on the resistance of Japanese pears to black spot disease. l. Difference of resistance among varieties and toxic substance produced by the causal fungus. J Hortic Assoc Japan 1947;16:181-91.
86. Hiroe I, Aoe S. Phytopatholog Japanese pear, caused by Alternaria kikuchiana Tanaka (English Series 1). Pathochemical studies (I). On a new phytotoxin, phyto-alternarin produced by the fungus. J Fac Agric Tottori Univ 1954;2:1-28.
87. Hiroe I, Nishimura S, Sato M. Pathochemical studies on black spot disease of Japanese pear (Part I). On leaf necrotic toxins produced by the causal fungus (l). Trans. Tottori Soc Agric Sci 1958;11:291-9.
88. Mohri R. Studies on the resistance of Japanese pears to the black spot disease. Lib Arts J Tottori Univ 1962;13:53-119.
89. Ohkawa M, Torikata H. Studies on the resistance of Japanese pears to black spot disease fungus (Alternaria kikuchiana Tanaka). VI. The isolation of host-specific toxin produced by Alternaria kikuchiana Tanaka. J Japan Soc Hortic Sci 1967;36:141-6. [CrossRef]
90. Prieto KR, de Medeiros LS, Isidoro MM, Toffano L, da Silva MF, Fernandes JB, et al. Rapid detection of ACTG- and AK-toxins in Alternaria alternata by LC-ESI-MS/MS analysis and antifungal properties of Citrus compounds. J Braz Chem Soc 2016;27:1493-505. [CrossRef]
91. Otani H, Nishimura S, Kohmoto K. Nature of specific susceptibility to Alternaria kikuchianain Nijissieki cultivar among Japanese pears. J Fac Agric Totrori Univ 1972;7:5-12.
92. Otani H, Nishimura S, Kohmoto K. Nature of specific susceptibility to Alternaria kikuchiana in Nijisseiki cultivar among Japanese pears. II. Effect of host-specific toxin on permeability of pear leaves (supplementary report). Fac Agric Totrori Univ 1973;8:14-20.
93. Otani H, Nishimura S, Kohmoto K, Yano K, Seno T. Nature of specific susceptibility to Alternaria kikuchiana in Nijisseiki cultivar among Japanese pears. V. Role of host -specific toxin in early step of infection. Ann Phytopathol Soc Japan 1975;41:467-76. [CrossRef]
94. Nishimura S, Sugihara M, Kohmoto K, Otani H. Two different phases in pathogenicity of the Alternaria pathogen causing black spot disease of Japanese pear. J Fac Agric Totturi Univ 1978;13:1-10.
95. Gardner JM, Kono Y, Tatum JH, Suzuki Y, Takeuchi S. Structure of the major component of ACRL-toxins, host-specific pathotoxic compound produced by Alternaria citri. Agric Biol Chem 1985;49:1235-8. [CrossRef]
96. Akimitsu K, Kohmoto K, Otani H, Nishimura S. Host-specific effects of toxin from the rough lemon pathotype of Alternaria alternata on mitochondria. Plant Physiol 1989;89:925-31. [CrossRef]
97. Pedras MS, Zaharia IL, Gai Y, Smith KC, Ward DE. Metabolism of the host-selective toxins destruxin B and homodestruxin B:Probing a plant disease resistance trait. Org Lett 1999;1:1655-8. [CrossRef]
98. Nakatsuka S, Ueda K, Goto T, Yamamoto M, Nishimura S, Kohmoto K. Structure of AF-toxin II, one of the host-specific toxins produced by Alternaria alternata strawberry pathotype. Tetrahedron Lett 1986;27:2753-6. [CrossRef]
99. Lee HB, Kim CJ, Yu SH. First report of strawberry fruit rot caused by Alternaria tenuissima in Korea. Plant Dis 2001;85:563. [CrossRef]
100. Meena M, Zehra A, Dubey MK, Upadhyay RS. Mannitol and proline accumulation in Lycopersicum esculentum during infection of Alternaria alternataand its toxins. Int J Biomed Sci Bioinform 2016;3:64-8.
101. Nakashima T, Ueno T, Fukami H, Taga T, Masuda H, Osaki K. Isolation and structure of AK-toxin I and II, host specific phytotoxic metabolites produced by Alternaria alternata Japanese pear pathotype. Agric Biol Chem 1985;49:807-15. [CrossRef]
102. Izumi Y, Ohtani K, Miyamoto Y, Masunaka A, Fukumoto T, Gomi K, et al.Apolyketide synthase gene, ACRTS2, is responsible for biosynthesis of host-selective ACR-toxin in the rough lemon pathotype of Alternaria alternata. Mol Plant Microbe Interact 2012;25:1419-29. [CrossRef]
103. Bottini AT, Gilchrist DG, Phytotoxins I. A 1-aminodimethylhepta decapentol from Alternaria alternata f. sp. lycopersici. Tetrahedron Lett 1981;22:2719-22. [CrossRef]
104. Kohmoto K, Itoh Y, Shimomura N, Kondoh Y, Otani H, Kodama M. Isolation and biological activities of two host-specific toxins from the tangerine pathotype of Alternaria alternata. Phytopathology 1993;83:495-502. [CrossRef]
105. Abbas HK, Tanaka T, Duke SO, Porter JK, Wray EM, Hodges L, et al. Fumonisin- and AAL-toxin-induced disruption of sphingolipid metabolism with accumulation of free sphingoid bases. Plant Physiol 1994;106:1085-93. [CrossRef]
106. Quayyum HA, Gijzen M, Traquair JA. Purification of a necrosis-inducing, host-specific protein toxin from spore germination fluid of Alternaria panax. Phytopathology 2003;93:323-8. [CrossRef]
107. Todd RB. Andrianopoulos A. Evolution of a fungal regulatory gene family:The Zn(II)Cys6 binuclear cluster DNA binding motif. Fungal Genet Biol 1997;21:388-405. [CrossRef]
108. Chaube HS, Pundhir VS. Crop Diseases and their Management. New Jersey:Prentice-Hall of India Private Limited;2005.
109. Tsuge T, Harimoto Y, Akimitsu K, Ohtani K, Kodama M, Akagi Y, et al. Host-selective toxins produced by the plant pathogenic fungus Alternaria alternata. FEMS Microbiol Rev 2013;37:44-66. [CrossRef]
110. Takaoka S, Kurata M, Harimoto Y, Hatta R, Yamamoto M, Akimitsu K, et al. Complex regulation of secondary metabolism controlling pathogenicity in the phytopathogenic fungus Alternaria alternata. New Phytol 2014;202:1297-309. [CrossRef]
111. Meena M, Swapnil P, Upadhyay RS. Isolation, characterization and toxicological potential of tenuazonic acid, alternariol and alternariol monomethyl ether produced by Alternaria species phytopathogenic on plants. Sci Rep 2017a;7:8777. [CrossRef]
112. Meena M, Gupta SK, Swapnil P, Zehra A, Dubey MK, Upadhyay RS. Alternaria toxins:Potential virulence factors and genes related to pathogenesis. Front Microbiol 2017b;8:1451. [CrossRef]
113. Liakopoulou-Kyriakides M, Lagopodi AL, Thanassoulopoulos CC, Stavropoulos GS, Magafa V. Isolation and synthesis of a host-selective toxin produced by Alternaria alternata. Phytochemistry 1997;45:37-40. [CrossRef]
114. Ma YT, Qiao LR, Shi WQ, Zhang AL, Gao JM. Metabolites produced by an endophyte Alternaria alternata isolated from Maytenus hookeri. Chem Nat Compd 2010;46:504-6. [CrossRef]
115. Bains PS, Tewari JP. Purification, chemical characterization and host-specificity of the toxin produced by Alternaria brassicae. Physiol Mol Plant Pathol 1987;30:259-371. [CrossRef]
116. Stierle AC, Cardellina JH, Strobel GA. Maculosin, a host-specific phytotoxin for spotted knapweed from Alternaria alternata. Proc Natl Acad Sci U S A 1988;85:8008-11. [CrossRef]
117. Stierle AC, Cardellina II, Strobel GA. Phytotoxins from Alternaria alternata, a pathogen of spotted knapweed. J Nat Prod 1989;52:42-7. [CrossRef]
118. Taj G, Meena PD, Giri P, Pandey D, Kumar A, Kumar A. Pathogenesis mechanisms employed by Alternaria species. J Oilseed Brassica 2015;6:213-40.
119. Iwamoto K, Takamatsu S, Yamamoto M. Alternaria alternata causing black spot of peach produces a host-specific toxin. J Gen Plant Pathol 2019;85:395-400. [CrossRef]
120. Kohmoto K, Otani H, Tsuge T. Alternaria alternata pathogens. In:Singh US, Singh RP, editors. (Pathogenesis and Host-Specificity in Plant Disease:Histopathological Biochemical, Genetic and Molecular Bases. Eukaryotes). Vol. 2. Oxford:Pergamon Press;1995. 51-63.
121. Meena M, Samal S. Alternaria host-specific (HSTs) toxins:An overview of chemical characterization, target sites, regulation and their toxic effects. Toxicol Rep 2019;6:745-58. [CrossRef]
122. Kohmoto K, Otani H, Nishimura S. Primary action sites for hostspecific toxins produced by Alternaria species. In:Nishimura S, editor. Molecular Determinants of Plant Diseases. Tokyo, Berlin:Springer-Verlag:Japan Scientific Societies Press;1987. 127-43.
123. Ueno T. Secondary metabolites related to host selection by plant pathogenic fungi. Pure Appl Chem 1990;62:1347-52. [CrossRef]
124. Arny DC, Nelson RR. Phyllosticta maydisspecies nova, the incitant of yellow leaf blight of maiza. Phytopathology 1971;61:1170-2. [CrossRef]
125. Mukunya DM, Boothroyd CW. Mycosphaerella zeae-maydis, the sexual stage of Phyllosticta maydis. Phytopathology 1973;63:529-32. [CrossRef]
126. Dewey RE, Siedow JN, Timothy CS, Levings CS 3rd. A 13-kilodalton maize mitochondrial protein in E. coli confers sensitivity to Bipolaris maydis toxin. Sci 1988;239:293-5. [CrossRef]
127. Condon BJ, Elliott C, González JB, Yun SH, Akagi Y, Wiesner-Hanks T, et al. Clues to an evolutionary mystery:The genes for T-Toxin, enabler of the devastating 1970 southern corn leaf blight epidemic, are present in ancestral species, suggesting an ancient origin. Mol Plant Microbe Interact 2018;31:1154-65. [CrossRef]
128. Danko S, Kono Y, Daly JM, Suzuki Y, Takeuchi S, McCrery DA. Structural and biological activity of a host-specific toxin produced by the fungal corn pathogen Phyllosticta maydis. Biochemistry 1984;23:759-66. [CrossRef]
129. Yoder OC. A selective toxin produced by Phyllosticta maydis. Phytopathology 1973;63:1361-5. [CrossRef]
130. Suzuki Y, Danko SJ, Kono Y, Daly JM, Knoche HW, Takeuchi S. Studies on the confirmations of PM-toxin, the host-specific corn pathotoxin produced by Phyllosticta maydis. Agric Biol Chem 1988;52:15-24. [CrossRef]
131. Johnson J, Murwin HF. Experiments on the control of wildfire tobacco. Wisconsin Agric Exp Stat Res Bull 1925;62:35.
132. Clayton EE. Toxin produced by Bacterium tabacum and its relation to host range. J Agric Res 1934;48:411-26.
133. Myung IS, Kim JW, An SH, Lee JH, Kim SK, Lee YK, et al. Wildfire of soybean caused by Pseudomonas syringae pv. tabaci, a new disease in Korea. Plant Dis 2009;93:1214. [CrossRef]
134. Yang P, Zhao L, Gao YG, Xia Y. Detection, diagnosis, and preventive management of the bacterial plant pathogen Pseudomonas syringae. Plants (Basel) 2023;12:1765. [CrossRef]
135. Lydon J, Patterson CD. Detection of tabtoxin-producing strains of Pseudomonas syringae by PCR. Lett Appl Microbiol 2001;32:166-70. [CrossRef]
136. Mapuranga N. A new race of Pseudomonas syringae pv. tabaci on tobacco in Zimbabwe. Plant Dis 1998;82:1404. [CrossRef]
137. Wooley DW, Pringle RB, Braun AC. Isolation of the phytopathogenic toxin of Pseudomonas tabaci, an antagonist of the methionine. J Biol Chem 1952;197:409-17. [CrossRef]
138. Stewart WW. Isolation and proof of structure of wildfire toxin. Nature 1971;229:174-8. [CrossRef]
139. Taylor PA, Schnoes HK, Durbin RD. Characterization of chlorosis-inducing toxins from a plant pathogenic Pseudomonas Sp. Biochim Biophys Acta 1972;286:107-17. [CrossRef]
140. Sinden SL, Durbin RD. Glutamine synthetase inhibition:Possible mode of action of wildfire toxin from Pseudomonas tabaci. Nature 1968;219:379-80. [CrossRef]
141. González-Villanueva L, Arvizu-Gómez JL, Hernández-Morales A, Aguilera-Aguirre S, Álvarez-Morales A. The PhtL protein of Pseudomonas syringae pv. phaseolicola NPS3121 affects the expression of both phaseolotoxin cluster (Pht) and Non-Pht encoded genes. Microbiol Res 2014;169:221-31. [CrossRef]
142. Sherf AF. Vegetable Diseases and their Control. United States:Wiley &Sons;1986. 36-41.
143. Sawada H, Fujikawa T. Genetic diversity of Pseudomonas syringae pv. actinidiae, pathogen of kiwifruit bacterial canker. Plant Pathol 2019;68:1235-48. [CrossRef]
144. Schwartz HF. Bacterial Diseases of Beans. Vol. 2. Colorado:Colorado State University Extension;2013. 913.
145. Bachmann AS, Matile P, Slusarenko AJ. Inhibition of ornithine decarboxylase activity by phaseolotoxin:Implications for symptom production in halo blight of French bean. Physiol Mol Plant Pathol 1998;53:287-99. [CrossRef]
146. Templeton GE, Grable CI, Fulton ND, Meyer WL. Tentoxin from Alternaria tenuis:Its Isolation and Characterization. In:Proceedings of the Mycotoxin Research Seminar, Washington, D.C, United States Department of Agriculture;1967. p. 27-9.
147. Lax AR, Shepherd HS, Edwards JV. Tentoxin, a chlorosis-inducing toxin from Alternaria as a potential herbicide. Weed Technol 1988;2:540-4. [CrossRef]
148. Vaughn KC, Duke S. Tentoxin stops the processing of polyphenol oxidase into an active protein. Physiol Plant 1984;60:257-61. [CrossRef]
149. Taylor TV, Mitchell TK, Daub ME. An oxidoreductase is involved in cercosporin degradation by the bacterium Xanthomonas campestris pv. zinniae. Appl Environ Microbiol 2006;72:6070-8. [CrossRef]
150. Valenzeno DP, Pooler JP. Photodynamic action. BioScience 1987;37:270-5. [CrossRef]
151. Bilski P, Li MY, Ehrenshaft M, Daub ME, Chignell CF. Vitamin B6 (pyridoxine) and its derivatives are efficient singlet oxygen quenchers and potential fungal antioxidants. Photochem Photobiol 2000;71:129-34. [CrossRef]
152. Daub ME, Chung KR. Cercosporin:A Photoactivated Toxin in Plant Disease. Online. United States:APSnet Features;2007. [CrossRef]
153. Chung KR. Involvement of calcium/calmodulin signaling in cercosporin toxin biosynthesis by Cercospora nicotianae. Appl Environ Microbiol 2003;69:1187-96. [CrossRef]
154. Shim WB, Dunkle LD. CZK3, a MAP kinase kinase kinase homolog in Cercospora zeae-maydis, regulates cercosporin biosynthesis, fungal development, and pathogenesis. Mol Plant Microbe Interact 2003;16:760-8. [CrossRef]
155. Chung KR, Daub ME, Kuchler K, Schuller C. The CRG1 gene required for resistance to the singlet oxygen-generating cercosporin toxin in Cercospora nicotianae encodes a putative fungal transcription factor. Biochem Biophys Res Commun 2003;302:302-10. [CrossRef]
156. Hellmers E. Bacterial leaf spot of African marigold (Tagetes erecta) caused by Pseudomonas tagetis sp. n. Acta Agric Scand 1955;5:185-200. [CrossRef]
157. Gulya TJ, Urs R, Banttari EE. Apical chlorosis of sunflower caused by Pseudomonas syringaepv. tagetis. Plant Dis 1981;66:598-600. [CrossRef]
158. Styer DJ, Durbin RD. Common ragweed:A new host of Pseudomonas syringae pv. tagetis. Plant Dis 1982;66:71. [CrossRef]
159. Shane WW, Baumer JS. Apical chlorosis and leaf spot of Jerusalem artichoke incited by Pseudomonas syringae pv. tagetis. Plant Dis 1984;68:257-60. [CrossRef]
160. Rhodehamel NH, Durbin RD. Host range of strains of Pseudomonas syrinage pv. tagetis. Plant Dis 1985;69:589-91. [CrossRef]
161. Rhodehamel NH, Durbin RD. Two new hosts of Pseudomonas syringae pv. tagetis. Plant Dis 1989a;73:368. [CrossRef]
162. Mitchell RE, Durbin RD. The structure of tagetitoxin, a phytotoxin of Pseudomonas syringae pv. tagetis. Phytochemistry 1983;22:1425-8. [CrossRef]
163. Mitchell RE, Coddington JM, Young H. A revised structure for tagetitoxin. Tetrahedron Lett 1989;30:501-4. [CrossRef]
164. Mathews DE, Durbin RD. Tagetitoxin inhibits RNA synthesis directed by RNA polymerases from chloroplasts and Escherichia coli. J Biol Chem 1990;265:493-4. [CrossRef]
165. Steinberg TH, Mathews DE, Durbin RD, Burgess RR. Tagetitoxin:A new inhibitor of eukaryotic transcription by RNA polymerase III. J Biol Chem 1990;265:499-505. [CrossRef]
166. Rhodehamel NH, Durbin RD. Toxin production by strains of Pseudomonas syringae pv. tagetis. Physiol Mol Plant Pathol 1989b;35:301-11. [CrossRef]
167. Farr DF, Castlebury LA, Pardo-Schultheiss RA. Phomopsis amygdali causes peach shoot blight of cultivated peach trees in the southeastern United States. Mycologia 1999;91:1008-15. [CrossRef]
168. Turner NC, Graniti A. Fusicoccin:A fungal toxin that opens stomata. Nature 1969;223:1070-1. [CrossRef]
169. Ballio A, Brufani M, Casinovi CG, Cerrini S, Fedeli W, Pellicciari R, et al. The structure of fusicoccin A. Experientia 1968;24:631-5. [CrossRef]
170. Barrow KD, Chain EB. The wilting toxins of Fusicoccum amygdali Del. Biochem J 1969;114:4P-5. [CrossRef]
171. Procacci R, Graniti A. Proceeding First Congress. Mediterranean Phytopathol Union, Bari;1966. 87.
172. Šišic A, Bacanovic-Šišic J, Al-Hatmi AM, Karlovsky P, Ahmed SA, Maier W, et al. The 'forma specialis'issue in Fusarium:A case study in Fusarium solani f. sp. pisi. Sci Rep 2018;8:1252.
173. Pfiffner A. Lsolierung und Konstitutionermittlung von Marticin und Isomarticin, Zwei Neuen Welketoxinen aus Fusarium Martii. Dissert. No. 3361. Zurlch:ETH;1963.
174. Singh RS. Introduction to Principles of Plant Pathology. 4th ed. New Delhi, India:Oxford and IBH Publishing Co. Pvt. Ltd.;2009. 161.
175. Kern H. Phytotoxins produced by Fusaria. In:Wood RK, Ballio A, Graniti A, editors. Phytotoxins in Plant Diseases. London, New York:Academic Press;1972. 35-45.
176. Gaumann E, Naef-Roth S, Kobel H. About Fusarinasure, a second wilt toxin from Fusarium lycopersici Sacc. Ibid 1952;20:1-38.
177. Gaumann E, Stoll C, Korn H. About Vasinfuscarin, a third Fusarium lycopersici Sacc. wilt toxin. Ibid 1953;20:245-47.
178. Woolley DW. Studies on the structure of lycomarasmin. J Biol Chem 1948;176:1291-8. [CrossRef]
179. Waggoner PE, Dimond AE. Role of chelation in causing and inhibiting the toxicity of lycomarasmine. Phytopathology 1953;43:281.
180. Ludwig RA. In:Horsfall JG, Dimond AE, editors. Plant Pathology. 2nd ed. New York:Academic Press;1960. 315.
181. Gaumann E. Fusaric acid as a wilt toxin. Phytopathology 1957;47:342-57.
182. Heitefuss R, Stahmann MA, Walker JC. Production of pectolytic enzymes and fusaric acid by Fusarium oxysporum, F. conglutinans in relation to cabbage yellows. Phytopathology 1960a;50:367-70.
183. Heitefuss R, Stahmann MA, Walker JC. Oxidative enzymes in cabbage infected by Fusarium oxysporum f. sp. conglutinans. Phytopathology 1960b;50:370-5.
184. Trione EJ. Extracellular enzyme and toxin production by Fusarium oxysporum f. sp. lini. Phytopathology 1960a;50:480-2.
185. Trione EJ. The HCN content of flax in relation to flax wilt resistance. Phytopathology 1960b;50:482-6.
186. Sandhu RS. Studies on the biogenesis of fusaric acid. Phytopathology 1960;37:33-60. [CrossRef]
187. Bohni N, Hofstetter V, Gindro K, Buyck B, Schumpp O, Bertrand S, et al. Production of fusaric acid by Fusarium spp. in pure culture and in solid medium Co-cultures. Molecules 2016;21:370. [CrossRef]
188. Prasad M, Chaudhary SK. In vitro production of fusaric acid and its impact on growth and sporulation in Fusarium oxysporum f.sp. udum. Phytopathology 1974;80:279-82. [CrossRef]
189. Kalyanasundaram R, Venkata Ram CS. Production and systemic translocation of fusaric acid in. Fusarium infected cotton plants. J Indian Bot Soc 1956;35:7-10.
190. Kalyanasundaram R. Production of fusaric acid by Fusarium lycopersici Sacc. in the rhizosphere of tomato plants. Phytopathology 1958;32:25-34. [CrossRef]
191. Wang R, Huang J, Liang A, Wang Y, Mur LAJ, Wang M, et al. Zinc and copper enhance cucumber tolerance to fusaric acid by mediating its distribution and toxicity and modifying the antioxidant system. Int J Mol Sci 2020;21:3370. [CrossRef]
192. Aziiba AE, Chai Q, Coulter JA. Rice blast:A disease with implications for global food security. Agronomy 2019;9:451. [CrossRef]
193. Bilgrami KS, Verma RN. Fungal metabolites. In:Physiology of Fungi. New Delhi:Vikas Publishing House Pvt. Ltd.;1981. 421.
194. Lebrun MH, Orcival J, Duchrtic C. Resistance of rice to tenuazonic acid, a toxin from Pyricularia oryzae. Revue Cytol Bioi Veg Bot 1984;7:249.
195. Sanmathi Kumar RP, Shanthala L, Anilkumar TB, Sudharshana L. Phytotoxins from Pyricularia grisea and their effect on finger millet. J Plant Biochem Biotechnol 2006;15:63-6. [CrossRef]
196. Nakajima T, Tamari K, Matsuda K. A cell wall proteo-heteroglycan from Piricularia oryzae: Isolation and partial structure. J Biochem 1977;82:1647-55. [CrossRef]
197. Koriem KM, Soliman RE. Chlorogenic and caftaric acids in liver toxicity and oxidative stress induced by methamphetamine. J Toxicol 2014;2014:583494. [CrossRef]
198. Mehrotra RS, Aggarwal A. Enzymes and toxins in plant diseases. In:Plant Pathology. New Delhi:Tata McGraw-Hill Publishing Company Ltd.;2008. 90-4.
199. Mugao L. Morphological and molecular variability of Alternaria solani and Phytophthora infestans causing tomato blights. Int J Microbiol 2023;2023:8951351. [CrossRef]
200. Singh RP. Colonization (Invasion and Establishment)-I:Attack by the pathogen on the host. In:Plant Pathology. 2nd ed. New Delhi:Kalyani Publishers;2012. 68-72.
201. Damm U, O'Connell RJ, Groenewald JZ, Crous PW. The Colletotrichum destructivumspecies complex - hemibiotrophic pathogens of forage and field crops. Stud Mycol 2014;79:49-84. [CrossRef]
202. McCarroll DR, Thor E. Do “toxins”affect pathogenesis by Endothia parasitica?Physiol Plant Pathol 1985;26:357-66. [CrossRef]
203. Apoga D, Åkesson H, Jansson HB, Odham G. Relationship between production of the phytotoxin prehelminthosporol and virulence in isolates of the plant pathogenic fungus Bipolaris sorokiniana. Eur J Plant Pathol 2002;108:519-26. [CrossRef]
204. Miyazaki S, Jiang K, Kobayashi M, Asami T, Nakajima M. Helminthosporic acid functions as an agonist for gibberellin receptor. Biosci Biotechnol Biochem 2017;81:2152-9. [CrossRef]
205. Chavan SS, Sawant K, Aspak, Kumar S, Shukla M, Rana M, et al. Brown spot:An embryonic disease of rice. Agrica 2022;11:172-81. [CrossRef]
206. Atallah O, Yassin S. Aspergillus spp. eliminate Sclerotinia sclerotiorum by imbalancing the ambient oxalic acid concentration and parasitizing its sclerotia. Environ Microbiol 2020;22:5265-79. [CrossRef]
207. Martin-Dominguez V, Cabrera PI, Eidt L, Pruesse U, Kuenz A, Ladero M, et al. Production of fumaric acid by Rhizopus arrhizus NRRL 1526:A simple production medium and the kinetic modelling of the bioprocess. Fermentation 2022;8:64. [CrossRef]
208. Kuenz A, Eidt L, Prüße U. Biotechnological production of fumaric acid by Rhizopus arrhizus-reaching industrially relevant final titers. Fermentation 2023;9:588. [CrossRef]
209. Michelsen CF, Stougaard P. Hydrogen cyanide synthesis and antifungal activity of the biocontrol strain Pseudomonas fluorescens In5 from Greenland is highly dependent on growth medium. Can J Microbiol 2012;58:381-90. [CrossRef]
210. Li Y, Liu J, Adekunle D, Bown L, Tahlan K, Bignell DRD. TxtH is a key component of the thaxtomin biosynthetic machinery in the potato common scab pathogen Streptomyces scabies. Mol Plant Pathol 2019;20:1379-93. [CrossRef]
211. Hudec C, Biessy A, Novinscak A, St-Onge R, Lamarre S, Blom J, et al. Comparative genomics of potato common scab-causing Streptomyces spp. Displaying varying virulence. Front Microbiol 2021;12:716522. [CrossRef]
212. Royer M, Costet L, Vivien E, Bes M, Cousin A, Damais A, et al. Albicidin pathotoxin produced by Xanthomonas albilineans is encoded by three large PKS and NRPS genes present in a gene cluster also containing several putative modifying, regulatory, and resistance genes. Mol Plant Microbe Interact 2004;17:414-27. [CrossRef]
213. Zhao JY, Chen J, Shi Y, Fu HY, Huang MT, Rott PC, et al. Sugarcane responses to two strains of Xanthomonas albilineans differing in pathogenicity through a differential modulation of salicylic acid and reactive oxygen species. Front Plant Sci 2022;13:1087525. [CrossRef]
214. Fukuchi N, Isogai A, Nakayama J, Takayama S, Yamashita S, Suyama K, et al. Structure and stereochemistry of three phytotoxins, syringomycin, syringotoxin and syringostatin, produced by Pseudomonas syringae pv. syringae. J Chem Soc Perkin Trans 1992;1:1149-57. [CrossRef]
215. Bender CL, Alarcón-Chaidez F, Gross DC. Pseudomonas syringae phytotoxins:Mode of action, regulation, and biosynthesis by peptide and polyketide synthetases. Microbiol Mol Biol Rev 1999;63:266-92. [CrossRef]
216. Lee S, Cheon W, Kwon HT, Lee Y, Kim J, Balaraju K, et al. Identification and characterization of Pseudomonas syringaepv. syringae, a causative bacterium of apple canker in Korea. Plant Pathol J 2023;39:88-107. [CrossRef]
217. Cuppels DA, Ainsworth T. Molecular and physiological characterization of Pseudomonas syringae pv. tomato and Pseudomonas syringae pv. maculicola strains that produce the phytotoxin coronatine. Appl Environ Microbiol 1995;61:3530-6. [CrossRef]
218. Xin XF, He SY. Pseudomonas syringae pv. tomato DC3000:A model pathogen for probing disease susceptibility and hormone signaling in plants. Annu Rev Phytopathol 2013;51:473-98. [CrossRef]
219. Wang Z, Luo W, Cheng S, Zhang H, Zong J, Zhang Z. Ralstonia solanacearum -A soil borne hidden enemy of plants:Research development in management strategies, their action mechanism and challenges. Front Plant Sci 2023;14:1141902. [CrossRef]
220. Bennett RA, Billing E. Origin of the polysaccharide component of ooze from plants infected with Erwinia amylovora. J Gen Microbiol 1980;116:341-9. [CrossRef]
221. Singh P, Bugiani R, Cavanni P, Nakajima H, Kodama M, Otani H, et al. Purification and biological characterization of host-specific SV-toxins from Stemphylium vesicarium causing brown spot of European pear. Phytopathology 1999;89:947-53. [CrossRef]
222. Tanahashi M, Okuda S, Miyazaki E, Parada RY, Ishihara A, Otani H, et al. Production of host-selective SV-toxins by Stemphylium sp. causing brown spot of European pear in Japan. J Phytopathol 2017;165:189-94. [CrossRef]
223. Pedras MS, Biesenthal CJ. Vital staining of plant cell suspension cultures:Evaluation of the phytotoxic activity of the phytotoxins phomalide and destruxin B. Plant Cell Rep 2000;19:1135-8. [CrossRef]
224. Chen X, Hao X, Akhberdi O, Zhu X. Genomic and transcriptomic survey provides insights into molecular basis of pathogenicity of the sunflower pathogen Phoma macdonaldii. J Fungi (Basel) 2023;9:520. [CrossRef]