ARTICLE HIGHLIGHTS
Response surface methodology was used to optimize enzyme-assisted extraction conditions for Gamma-aminobutyric acid and polyphenols.
The conditions significantly affected the Gamma-aminobutyric acid and polyphenols contents of the mung bean extract.
The maximum content of Gamma-aminobutyric acid and polyphenols could be obtained using green and environmental-friendly techniques.
1. INTRODUCTION
Beans are known to synthesize several bioactive compounds that possess nutritional value during the germination process. According to Huang et al., [1] germination is a cost-effective and efficient process that modifies the physical properties of the beans and synthesizes bioactive compounds that have a positive impact on human health. Moreover, it breaks down high-molecular-weight compounds into simpler molecules that are more nutritious and easier for the human body to absorb. During this process, cellular components undergo transformations, and new compounds are formed that affect the biochemical properties of the beans. It enhances their quality by activating enzyme activity in germinated beans and converting proteins, carbohydrates, and lipids into simpler forms [2]. Germinated mung beans, for instance, exhibit an increase the nutritional content and improve protein digestibility, while also accumulating various bioactive compounds such as gamma-aminobutyric acid (GABA) and polyphenols [3], among others. Importantly, unwanted components such as alkaloids, tannins, and phytates are reduced during the germination process [4].
GABA is a non-protein amino acid containing four carbon atoms and having a molecular formula of C4H9NO2. It is synthesized from glutamic acid in the cytoplasm by a non-reversible reaction that removes the carboxyl group and releases CO2 through the specific enzyme glutamate decarboxylase in secretory cells when stimulated externally. GABA is found in various vegetables and germinated seeds such as grains, beans, soybeans, brown rice, sesame seeds, lentils, and peas [5-7]. According to Sarasa et al. [8], GABA plays a significant role as an inhibitory neurotransmitter that suppresses the transmission of nerve impulses, regulates blood pressure, controls stress responses, and modulates sleep, and is one of the ingredients in tranquilizers. GABA also plays a vital role in promoting human health by reducing nervous tension, improving sleep quality, alleviating symptoms of nervous disorders, and stimulating immune cell production.
Similarly, it is reported that polyphenols in various seeds, such as germinated rice, increased during germination [9]. Plant phenolic compounds have the ability to protect against ultraviolet radiation or prevent disease-causing agents, as well as enhance the color of plants. Due to their presence in all parts of the plant, these compounds are integral components of the human diet. In addition, polyphenols act as anti-mutagenic, anti-allergic, anti-inflammatory, and antibacterial agents [10]. Polyphenols have antioxidant activity and can prevent cardiovascular diseases through their antioxidative mechanisms and by reducing the impact of harmful cholesterol [11]. According to Avinash et al. [12], extracts derived from Atibala (Abutilon indicum) and its polyphenolic compounds exhibit strong inhibitory effects on Pseudomonas aeruginosa biofilm. These results hold promise for their potential utilization in the future for the design and development of innovative drugs.
The successful extraction of bioactive compounds from plant materials, especially germinated mung beans, strongly depends on the appropriate selection of extraction methods. Modern techniques such as ultrasound-assisted extraction, microwave-assisted extraction, and enzyme-assisted extraction (EAE) offer notable advantages over conventional methods such as maceration, percolation, and Soxhlet extraction. As a result, these advanced methods find wider applications in the food, cosmetics, and pharmaceutical industries [13]. The choice of suitable extraction methods depends on the nature of the raw material, the preparation process, and energy consumption. It is necessary to choose a method that minimizes the loss of natural bioactive compounds, consumes less energy, yields the highest amount of bioactive compounds, and is environmentally friendly. Among them, EAE stands out as an environmentally friendly method, as it not only reduces extraction time but also enhances extraction efficiency and cost-effectiveness. Moreover, EAE is known for its gentle extraction technology, making it a highly promising alternative to conventional extraction methods [14]. EAE has found extensive application in obtaining bioactive compounds from a range of plant sources, such as flavonoids (naringin) from citrus peels [15,16], total phenolics from pomegranate peels [17], and bioactive peptides with high antioxidant activity from okara protein [18].
EAE mainly relies on the specific catalytic ability of enzymes and the catalytic reaction occurring under mild conditions in an aqueous environment [19]. Enzymes are used in the extraction process because they break down the cell structure of plant material, thereby significantly improving the efficiency of extracting bioactive compounds from plants. Through enzyme hydrolysis, the cell walls are broken down, leading to enhanced cell permeability and enabling the extraction of bioactive compounds in substantial yields. The selection of enzymes is contingent upon the intrinsic characteristics of individual source materials and the desired constituents. For instance, previous studies have demonstrated the use of cellulase enzymes for extracting bioactive compounds from mung bean skin [20], while enzymes from the protease group, such as alcalase and papain, have been employed for extracting proteins and amino acids from seeds [21]. Since GABA represents an amino acid from which phenolic compounds are synthesized, alcalase has proven to be the leading enzyme selected for extracting bioactive compounds from germinated mung beans. To effectively use enzymes for the extraction process, a comprehensive understanding of the enzyme’s catalytic properties, mode of action, and the ideal conditions of the enzyme that best suit the chosen plant material is essential. The necessary parameters for each individual process related to the release of bioactive compounds with enzyme assistance include material-to-water ratio, temperature, time, and enzyme concentration [16]. Moreover, it is necessary to investigate the cell composition of the raw material before selecting an appropriate enzyme or combination of enzymes [22]. Therefore, the EAE process requires a clear understanding of the nature of the raw material, the selection of enzymes for different purposes, and the establishment of optimal conditions for the extraction process.
To determine the parameters of the EAE process and optimize the factors affecting it, such as raw material-water ratio, extraction temperature, extraction time, and enzyme concentration during extraction, the research investigated the factors that affect the extraction process and optimized these factors to achieve the utmost amount of bioactive compounds in the resultant extract using alcalase enzyme.
2. MATERIALS AND METHODS
2.1. Materials
For this study, alcalase 2.5 L PF with an activity of 2.5 AU-A/g was acquired from Novozymes (Denmark). All chemicals and reagents used, including ethanol, sodium hypochlorite, sodium carbonate anhydrous, phenol, boric acid, and Folin–Ciocalteu, were of analytical grade and obtained from Merck (Germany). The standard substances, namely GABA and gallic acid, were procured from Sigma Aldrich (USA).
Mung beans of the DX208 variety were obtained from the Southern Seed Corporation, Vietnam. The beans were selected by removing broken beans, flat beans, and impurities and stored at 10–12°C in a refrigerator to prevent spoilage. They were soaked in water with a ratio of 1:5 (w/w) at the temperature of 30°C for 8 h [23]. After soaking, they were washed with water and spread out on a stainless steel tray in a 1 cm layer. The tray was then placed inside an incubator set at 35°C with 90% humidity for 12 h [24]. After incubation, the germinated mung beans were taken out of the incubator, and washed with distilled water to remove the mucilaginous substances that might have formed on the surface. Afterward, they were allowed to to drain for 10 min. Subsequently, they were packaged and stored at a temperature of −18°C to be utilized for further extraction experiments.
2.2. Preliminary Experiments to Determine the Suitable Conditions for EAE
Preliminary experiments were conducted to investigate the factors influencing the extraction of bioactive compounds from germinated mung beans. About 3 g of germinated mung beans, sourced from the stock kept at −18°C, were mixed with an appropriate amount of water (Section 2.2.1) and ground in a laboratory blender for 2 min. The resulting slurry was then adjusted to pH 8 using a 1M NaOH solution [21]. EAE was carried out under varying conditions, including the mung bean-to-water ratio, temperature, time, and enzyme concentration. After hydrolysis, the alcalase was inactivated by placing the solution in a heat bath at 95°C for 10 min. The resulting hydrolysate was then cooled, and centrifuged at 5000 rpm at 4°C for 15 min, and the upper portion of the solution was collected, filtered, and stored at a low temperature of 10–12°C for a short period of approximately 2 h for the analysis of GABA and polyphenol contents. The results of the previous experiment served as the basis for the subsequent experiments.
2.2.1. The effects of the ratio of mung bean to water
Different mung bean-to-solvent ratios (w/w) of 1:5, 1:10, 1:15, 1:20, and 1:25 were investigated. The fixed factors, including 1% (w/w) enzyme concentration and an incubation temperature of 40°C for 30 min, were used.
2.2.2. The effect of temperature
Hydrolysis was carried out by placing the bean solution in a water bath at temperatures of 40, 50, and 60°C for 30 min.
2.2.3. The effect of time
The investigated time of hydrolysis was 30, 60, and 90 min at the mung bean-to-water ratio and the temperature obtained from the previous experiments.
2.2.4. The effect of enzyme concentration
Different concentrations of alcalase of 1%, 2%, 3%, and 4% (w/w) were investigated. Enzymatic hydrolysis was conducted by placing the mung bean slurry in a water bath at the temperature for the duration determined from the previous experiments.
2.3. Optimization of Extraction Conditions
After identifying the independent factors in the preliminary experiments, the response surface methodology (RSM) was employed to optimize the extraction conditions. A Box-Behnken design (BBD) with 3 experimental factors, each at 3 different levels [Table 1], was used for this purpose. The investigated factors were extraction temperature, extraction time, and enzyme concentration. The efficiency of the extraction conditions of germinated mung beans was evaluated based on the GABA and polyphenols contents present in the extract.
Table 1: Code and uncoded levels for the three independent variables.
| Independent variable | Coded variable level | ||
|---|---|---|---|
| −1 | 0 | 1 | |
| Temperature (X1, °C) | 40 | 50 | 60 |
| Time (X2, min) | 40 | 60 | 80 |
| Enzyme concentration (X3, %) | 1 | 2 | 3 |
2.4. Analytical Methods
2.4.1. Determination of moisture content
Gravimetric analysis of the moisture content in germinated mung beans and their extracts was performed using the AnD MX-50 moisture analyzer (A and D Company, Ltd., Tokyo, Japan).
2.4.2. Determination of GABA
The procedure described by Watchararparpaiboon et al. [25], with slight modifications, was used to determine the GABA content in germinated mung beans through spectrophotometric analysis. To elaborate, 0.6 mL of the extracted solution was pipetted into a test tube, followed by the addition of 0.4 mL of 0.4 M borate buffer mixture and 2 mL of 6% phenol reagent. Then, 2 mL of 7.5% NaOCl reagent was added, and the tube was vigorously shaken for 5 min. The resulting mixture was incubated at 90°C for 10 min, followed by cooling on ice for another 10 min. The absorbance of the solution was measured at a wavelength of 630 nm using a spectrophotometer (Spectro UV 11, Germany). GABA served as a standard in this analysis and the results were expressed as milligrams (mg) of GABA per gram of dry weight (dw) of the sample (mg GABA/g, dw).
2.4.3. Determination of total polyphenol content (TPC)
To determine the TPC in the germinated mung bean extract, the modified method of Z?otek et al. [26] was used. The extracted solution (0.5 mL) was mixed with 2 mL of distilled water, followed by the addition of 0.5 mL of Folin–Ciocalteu reagent (at a ratio of 1:10, v/v) and 2 mL of 7.0% Na2CO3. The mixture was then thoroughly shaken and allowed to stand at room temperature for an hour. The absorbance of the solution was measured at 765 nm using a spectrophotometer (Spectro UV 11, Germany). The results were expressed as milligrams of gallic acid equivalents (GAE) per gram of dry weight (dw) of the sample (mg GAE/g, dw), with gallic acid being used as the standard.
2.5. Statistical Analyses
The experiments were performed in triplicate, and the measurements were also done in triplicate. Statistical analysis was performed using one-way analysis of variance (ANOVA), and the least significant difference post hoc test at a significance level of 5% (P < 0.05) to identify significant differences between mean values. SPSS software version 25.0 (IBM Corp., Armonk, NY, USA) was used for data analysis, and the mean values with error bars were plotted using Excel version 2016 (Microsoft, Seattle, WA, USA).
To analyze the content of GABA and TPC, quadratic regression equations were created using JMP software version 13.0 (SAS Institute Inc., Cary, NC, USA). Additionally, three-dimensional (3D) surface and two-dimensional (2D) contour plots were generated to visualize the relationship between the variables. The models’ adequacy was assessed based on the coefficient of determination (R2) and the lack of fit. A second-order polynomial equation was utilized to express the relationship between the content of GABA (Y1) and TPC (Y2) as a function of the independent variables. The equation was expressed as follows:
Yi = ao + a1X1 + a2X2 + a3X3 + a11X21 + a22X22 + a33X23 + a12X1X2 + a13X1X3 + a23X2X3
Where, Yi represents the response variables, ao is a constant, ai, aii, aij are the linear, quadratic and interactive coefficients, respectively. Xi, Xj are the levels of the independent variables.
By keeping all terms in the equation, even those that may not individually demonstrate statistical significane, the model maintains a consistent and cohesive representation of the data and the underlying interactions between the variables.
3. RESULTS AND DISCUSSION
3.1. Preliminary Experiments to Determine the Suitable Conditions for EAE
3.1.1. The effects of the ratio of mung bean to water
Water plays a crucial role in enzymatic hydrolysis, supporting the reaction and the dynamic nature of the enzyme and its products [22]. A low solid-liquid ratio leads to the formation of a highly viscous solution, making it difficult for the enzyme and substrate to come into contact. However, if the ratio of raw material and solvent is too high, it dilutes the product and increases liquid waste and emulsification, thereby increasing the cost of waste treatment and emulsion removal [27]. The ratio of raw material to water is not fixed and depends on the type and properties of the raw materials, typically ranging from 10 to 40 (w/w). For germinated mung beans, the ratio of raw material to water affects the content of bioactive compounds extracted, as shown in Figure 1.
 | Figure 1: The effect of the mung bean-to-water ratio on the content of gamma-aminobutyric acid (a) and total polyphenols (b). Values in a column not sharing a superscript letter (a, b, c, d, e) are significantly different from each other (P < 0.05). [Click here to view] |
According to statistical analysis, the influence of the mung bean-to-water ratio on the GABA and TPC contents was found to be significant (P < 0.001). Based on Figure 1, the mung bean-to-water ratio increased from 1:5 to 1:20, resulting in a rapid increase in the content of GABA and TPC, with the highest GABA content reaching 34.66 mg/g, dw [Figure 1a], and the highest TPC content reaching 3.41 mg GAE/g, dw [Figure 1b] at a ratio of 1:20. However, when the ratio was increased further, the content of these compounds tended to decrease. In the initial stage, increasing the amount of water facilitated the contact between the water and the mung beans, thereby increasing the release of bioactive compounds in germinated mung beans. In the later stage, the increased water amount resulted in product dilution, limiting the release of bioactive compounds. Therefore, the mung bean-to-water ratio of 1:20 was found to be the most suitable and was selected for further optimization experiments.
3.1.2. The effects of temperature
Temperature affects the activity of enzymes and increases the reaction rate to the optimal level. The temperature above the point where enzymes become denatured is similar to the inactivity of proteins and the degradation of bioactive compounds. Depending on the type of enzyme, the hydrolysis process occurs at different temperatures for different raw materials, but most studies have conducted extractions at temperatures ranging from 30 to 60°C, and the enzyme activity decreases at 80°C. High temperatures require more energy and result in low-value products. Choosing an appropriate extraction temperature makes the hydrolysis reaction faster and yields valuable products. The impact of temperature on the extraction process of bioactive compounds is demonstrated in Figure 2.
 | Figure 2: The effects of incubation temperature on on the content of gamma-aminobutyric acid (a) and total polyphenols (b). Values in a column not sharing a superscript letter (a, b, c) are significantly different from each other (P < 0.05). [Click here to view] |
Statistical analysis indicated that the effects of incubation temperature significantly influenced the quantity of GABA (P < 0.01) and TPC (P < 0.05). As depicted in Figure 2, when the incubation temperature increased from 40 to 50°C, the amount of GABA increased and quickly reached a value of 35.86 mg/g, dw. When the temperature further increased to 60°C, the amount of GABA tended to decrease. These findings align with previous reports that employed alcalase enzyme at moderate temperatures for protein extraction from soybeans [28] or oil palm leaves [29]. Therefore, a temperature of 50°C was selected as the central point for optimization experiments.
3.1.3. The effects of time
The degradation process of cellular constituents can be enhanced by prolonging the hydrolysis time, which varies depending on the solvent and raw material source. Depending on the type of enzyme and the specific mechanisms involved, the time required may differ. However, prolonged exposure leads to the oxidation of bioactive compounds [30]. The results align with the findings reported in previous research, which demonstrate that an extended hydrolysis time leads to a reduction in extraction yield attributed to the degradation of bioactive compounds [28,31]. Figure 3 demonstrates the GABA and TPC contents in the extract affected by the extraction durations of 30–90 min. According to statistical analysis, the influence of the extraction duration on the amount of GABA (P < 0.001) and TPC (P < 0.01) was found to be significant.
 | Figure 3: The effects the effects of incubation time on the content of gamma-aminobutyric acid (a) and total polyphenols (b). Values in a column not sharing a superscript letter (a, b, c) are significantly different from each other (P < 0.05). [Click here to view] |
As can be seen in Figure 3, the enzyme extraction duration increased from 30 to 60 min, the total amount of GABA and polyphenols increased rapidly and reached the highest values of 51.32 mg/g, dw and 4.21 mg GAE/g, dw, respectively. When the processing time was prolonged beyond the optimum, the amount of bioactive compounds tended to decrease. Since the time exceeded the optimal level, the enzyme activity decreased, and the amount of bioactive compounds could be destroyed. Therefore, the incubation time of 60 min was chosen for the subsequent optimization experiments.
3.1.4. The effects of enzyme concentration
The ratio of enzyme to substrate is a fundamental factor that significantly affects the cost of the enzyme and the economy of the process. In general, the typical range of enzyme concentration used in experiments is between 0.2% and 3.0% [32]. At high enzyme concentrations, the formation of emulsions may occur, making it difficult to release bioactive compounds. As such, it is desirable to determine the most suitable enzyme concentration to achieve the highest amount of bioactive compounds in the resultant extract. Figure 4 shows the amount of GABA and TPC impacted by the various enzyme concentrations. In addition, statistical analysis indicated that the enzyme concentration significantly affects the amount of GABA and TPC (P < 0.001).
 | Figure 4: The effects of enzyme concentration on the gamma-aminobutyric acid content (a) and total polyphenols (b). [Click here to view] |
Upon increasing the enzyme concentration to 1% and 2%, the highest quantities of GABA and TPC reached were 57.71 mg/g, dw and 4.95 mg GAE/g, dw, respectively. However, upon surpassing a certain threshold of enzyme concentration, the amount of bioactive compounds displayed a tendency to decrease. This phenomenon can be attributed to the excessive amount of enzyme that can cause an intense hydrolysis reaction and a subsequent increase in bioactive compounds. Nonetheless, beyond a certain threshold, the enzyme reaction can attain equilibrium, and the hydrolysis reaction may not continue, leading to no further enhancement in the bioactive compound content and potentially even causing a decline. According to Fu et al. [33], when all substrate molecules are fully bound to the enzyme’s active site, the extraction process achieves its maximum yield as bioactive compounds are released from the material. However, increasing the enzyme quantity beyond this point leads to an excess of enzymes without a corresponding increase in the extraction yield. Moreover, excessive enzyme concentrations not only waste resources but can also negatively impact the extraction process, possibly inhibiting or interfering with the enzymatic reactions necessary for the extraction of desired compounds. Consequently, an enzyme concentration of 2% was chosen for the subsequent optimization experiments.
3.2. Optimization of EAE Conditions
3.2.1. Fitting the model
The RSM employs mathematical and statistical techniques to optimize experimental processes while minimizing the number of trials required. Table 2 shows the findings of the experiments in terms of GABA and TPC contents. The ANOVA results determined that the second-order equation was a suitable representation of the experimental data, with high R2 coefficients of 0.97 and 0.95 for GABA and TPC, respectively. The predicted values closely matched the experimental data acquired from the RSM design [Table 2]. Moreover, the ANOVA results demonstrated that the two quadratic regression equations were highly significant (P < 0.01), but the lack of fit was non-significant (P > 0.05) in either model [Table 3]. These findings indicate that the quadratic regression models accurately describe the effect of the independent variables investigated, i.e., temperature, time, and enzyme concentration, on the content of GABA and TPC.
Table 2: Experimental (Exp.) and predicted (Pred.) values of the GABA and TPC obtained from the Box-Behnken design.
| Exp. run | X1 (°C) | X2 (min) | X3 (%) | GABA (mg/g, dw) | TPC (mg GAE/g, dw) | ||
|---|---|---|---|---|---|---|---|
| Temp. | Time | Enzyme Conc. | Exp. | Pred. | Exp. | Pred. | |
| 1 | 0 | 0 | 0 | 55.563 | 56.352 | 6.247 | 6.454 |
| 2 | −1 | −1 | 0 | 45.037 | 44.414 | 4.316 | 4.230 |
| 3 | 0 | −1 | −1 | 41.831 | 41.268 | 3.748 | 3.680 |
| 4 | 1 | 0 | 1 | 54.418 | 53.232 | 5.226 | 5.072 |
| 5 | 1 | −1 | 0 | 51.281 | 50.881 | 4.87 | 4.736 |
| 6 | 0 | 1 | −1 | 48.346 | 46.760 | 5.374 | 5.086 |
| 7 | 0 | −1 | 1 | 45.938 | 47.524 | 4.198 | 4.486 |
| 8 | 0 | 0 | 0 | 57.453 | 56.352 | 6.858 | 6.454 |
| 9 | −1 | 0 | 1 | 42.28 | 41.317 | 5.221 | 5.019 |
| 10 | −1 | 0 | −1 | 43.986 | 45.172 | 3.863 | 4.017 |
| 11 | 1 | 1 | 0 | 51.089 | 51.712 | 5.798 | 5.884 |
| 12 | −1 | 1 | 0 | 48.606 | 49.006 | 5.057 | 5.191 |
| 13 | 0 | 0 | 0 | 56.039 | 56.352 | 6.256 | 6.454 |
| 14 | 0 | 1 | 1 | 46.891 | 47.454 | 5.122 | 5.190 |
| 15 | 1 | 0 | −1 | 41.465 | 42.428 | 4.963 | 5.165 |
GABA: Gamma-aminobutyric acid, TPC: Total polyphenol content.
Table 3: Regression coefficients of the fitted quadratic equation and standard errors for GABA and TPC.
| Regression coefficienta | GABA (Y1) | TPC (Y2) | ||
|---|---|---|---|---|
| Regression coefficients | t ratio | Regression coefficients | t ratio | |
| a0 | 56.352*** | 59.71 | 6.454*** | 32.27 |
| Linear | ||||
| a1 | 2.293* | 3.97 | 0.3 | 2.45 |
| a2 | 1.356 | 2.35 | 0.527** | 4.31 |
| a3 | 1.737* | 3.01 | 0.227 | 1.86 |
| Quadratic | ||||
| a11 | −3.781** | −4.45 | −0.618* | −3.43 |
| a22 | −3.567** | −4.19 | −0.826** | −4.58 |
| a33 | −7.033*** | −8.27 | −1.018** | −5.64 |
| Interaction | ||||
| a12 | −0.940 | −1.15 | 0.047 | 0.27 |
| a13 | 3.665** | 4.48 | −0.274 | −1.58 |
| a23 | −1.391 | −1.70 | −0.176 | −1.01 |
| R2 | 0.967 | 0.946 | ||
| P-value of lack of fit | 0.209 | 0.546 | ||
* P<0.05,
** P<0.01,
*** P<0.001.
a a0 is a constant, ai, aii, aij are the linear, quadratic, and interactive coefficients of the second-order polynomial equation, respectively, GABA: Gamma-aminobutyric acid, TPC: Total polyphenol content.
As shown in Table 3, the significance of the quadratic polynomial model coefficients is presented, and they were evaluated using t ratios and P-values at a given significance level. A higher absolute t-ratio and a lower P-value signify a greater impact on the relevant variables [34]. The variables that had the greatest effect on GABA content were the quadratic terms of X3, X1, and X2, as well as the interaction terms of X1 and X3, followed by the linear terms of X1 and X2. The lack of fit of the established mathematical model was found to be non-significant (P > 0.05), indicating a good fit of the model to the experimental data. The R2 coefficient of 0.967 indicates that 96.7% of the GABA content can be explained by this model.
Regarding TPC content, the quadratic terms of enzyme concentration, time, and temperature were the most influential factors, while the linear term of time also made a significant contribution. In contrast, the interaction among the three independent variables had a non-significant effect on TPC content (P > 0.05). The lack of fit of the mathematical model developed to predict the TPC content was also non-significant (P > 0.05), and it had an R2 coefficient of 0.946, indicating that the model explains approximately 94.6% of the variation in TPC content.
3.2.2. Response surface optimization of GABA content
To assess the model’s fit and determine the ideal levels of the independent variables for the content of GABA in germinated mung bean extract, 3D surface and 2D contour plots were utilized. The response surface plot displays the relationship between the independent variables and the responses, providing a visual representation of how the content of GABA is affected by changes in temperature, time, and enzyme concentration. On the other hand, the contour plot aids in visualizing the shape of the response surface and helps identify regions of optimal response. Hence, these plots are useful tools for evaluating the model’s accuracy [35] and guiding the selection of the most favorable conditions to maximize the GABA content during the extraction process.
Table 3 presents the impact of temperature, time, and enzyme concentration on the content of GABA, as represented by the coefficients of the quadratic regression equation. The corresponding response surface and contour charts representing the content of GABA based on these coefficients are shown in Figure 5. In general, all regression coefficients of the equation had a statistically significant influence on the content of GABA, except for the linear term of X2 and the interaction terms of X1 and X2 and X2 and X3 (P > 0.05). Figure 5a indicates that increasing the incubation temperature from 40 to 54°C and the time from 40 to 62 min, with a constant enzyme concentration of 2%, led to a substantial increase in GABA content. However, the content of GABA gradually decreased with increasing temperature and time, up to 60°C and 80 min, respectively. Similarly, at a constant incubation time of 60 min [Figure 5b], the GABA content exhibited a similar trend with varying temperatures (40–54°C) and enzyme concentration (1–2.2%). As shown in Figure 5c, the content of GABA increased when time and enzyme concentration was increased at a constant incubation temperature of 50°C. The critical values of incubation temperature, time, and enzyme concentration for GABA extraction were 54°C, 62 min, and 2.2%, respectively. At this point, the GABA content was about 57.06 mg/g, dw. These findings provide valuable insights into the optimum conditions for maximizing the GABA content during the extraction process.
 | Figure 5: (a-c) The 3D response surface and 2D contour plots of gamma-aminobutyric acid and total polyphenol content affected by temperature (X1), time (X2), and enzyme concentration (X3). [Click here to view] |
3.2.3. Response surface optimization of TPC content
According to the statistical analysis, the linear term of X2 (P < 0.01) and quadratic terms of X1 (P < 0.05), X2 (P < 0.01), and X3 (P < 0.01) had a significant impact on the TPC content of the mung bean extract, as shown in Table 3. Figure 6a shows the interactions between temperature and time on the TPC content. Similar to the GABA, content trend is shown in Figure 5a, medium extraction parameters of incubation temperature and duration yielded the greater TPC content of 6.56 (mg GAE/g, dw). In addition, a high TPC content was obtained at the moderate temperature and enzyme concentration [Figure 6b], or at the medium incubation time and enzyme concentration [Figure 6c]. Furthermore, the statistical results revealed that the maximum TPC content (6.57 mg GAE/g, dw) could be obtained at the predicted extraction conditions, which included an incubation temperature, time, and enzyme concentration of 52°C, 65 min, and 2%, respectively.
 | Figure 6: (a-c) The 3D response surface and 2D contour plots of total polyphenol content affected by temperature (X1), time (X2), and enzyme concentration (X3). [Click here to view] |
The observed similar trend in the extraction conditions of the GABA and TPC contents can be attributed to the fact that these bioactive compounds are water soluble. As water is a major solvent used in the extraction process, it is likely that both GABA and TPC were efficiently extracted together with water under the investigated parameters. Additionally, the statistical analysis indicated that the quadratic terms of temperature, time, and enzyme concentration were significant independent variables that affected the content of GABA and TPC in both models. Specifically, under optimal parameters of incubation temperature and time, the optimal concentration of the alcalase enzyme played a crucial role in the extraction process. This can be explained by the fact that optimal enzyme concentration aids in breaking down the cell membrane, thereby enhancing the release of the bioactive compounds, leading to higher yields of GABA and TPC in the extract.
3.2.4. Overall optimal extraction conditions and model validation
The main objective of this work was to identify the optimal parameters for EAE of GABA and TPC contents and a graphical optimization was conducted using the JMP software program. Maximizing the contents of GABA and TPC was the primary criterion, and Figure 7 shows that increasing incubation temperature, time, and enzyme concentration led to higher contents of these bioactive compounds. However, further increasing these independent variables resulted in a decrease in the bioactive compounds. This is consistent with previous studies that have shown that these variables can positively influence the extraction of various bioactive compounds [36-38]. Moreover, the increasing trend of GABA and TPC contents showed some slight differences. As a result, determining the precise optimal parameters is crucial to achieving the maximum yield of GABA and TPC in the resultant extract. This information is essential for optimizing the extraction process and obtaining a high-quality extract rich in these valuable bioactive compounds from germinated mung beans.
 | Figure 7: Prediction profilers of gamma-aminobutyric acid and total polyphenol content as a function of temperature, time, and enzyme concentration. [Click here to view] |
The second-order polynomial equations for predicting the content of GABA (1) and TPC (2) according to the practical EAE conditions, including temperature (X1, °C), time (X2, min), and enzyme concentration (%) are as follows:
For GABA (Y1, mg/g, dw):
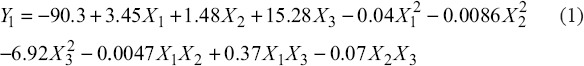 |
For TPC (Y2, mg GAE/g, dw):
 |
The prediction profiler from Figure 7 demonstrates that a combination of incubation temperature, time, and enzyme concentration could achieve the optimal content of GABA and TPC. The mathematical model used to predict the optimal theoretical values of GABA and TPC suggested that the optimal conditions for achieving these maximum values were a temperature of 53°C, a time of 62 min, and an enzyme concentration of 2.2%. To test the model’s accuracy, an experiment was run at the optimal parameters with three replicates, and the measured values were compared with the anticipated values. The results in Table 4 indicate that the measured values were in good agreement with the predicted values, thereby validating the quadratic regression equation for forecasting ideal response values under these conditions. Therefore, the response surface optimization for EAE conditions for GABA and TPC is of practical value.
Table 4: Optimal condition of enzyme-assisted extraction of GABA and TPC.
| Temperature (X1,°C) | Time (X2, min) | Enzyme concentration (X3, %) | GABA (mg/g, dw) | TPC (mg GAE/g, dw) | ||
|---|---|---|---|---|---|---|
| Predicted | Measured | Predicted | Measured | |||
| 53 | 62 | 2.2 | 57.01 | 57.73±1.28 | 6.52 | 6.81±0.42 |
GABA: Gamma-aminobutyric acid, TPC: Total polyphenol content.
The use of enzymes in extraction processes is widely recognized as beneficial due to their high efficiency, mild extraction conditions, shorter extraction times, and preservation of the properties and stability of the extract. The content of GABA and TPC in the resulting extracts [Figures 4 and 7] was significantly higher compared to extraction processes without enzyme assistance. Thus, the optimization process for GABA and TPC using EAE could encourage processors to utilize enzymes to enhance extraction yields. The established optimal conditions can be applied in practical applications to produce extracts enriched with these bioactive compounds for various potential health and nutritional benefits. In addition, a forthcoming paper will report on a comparison of different extraction methods for their effect on GABA and TPC content, polyphenol composition, and bioactive compound purification.
4. CONCLUSION
The EAE conditions for extracting bioactive compounds from germinated mung were successfully optimized using the RSM with BBD. Under the mung bean-to-water ratio of 1:20, quadratic regression equations were generated, which were statistically adequate in describing and predicting the responses for the GABA (R2 = 0.97) and TPC (R2 = 0.95) contents in the extract. The optimal conditions were determined to be an incubation temperature (X1) of 53°C, a time (X2) of 62 min, and an enzyme concentration (X3) of 2.2%. The content of GABA and TPC in the extract was predicted to be 57.01 mg/g, dw and 6.52 mg GAE/g, dw, respectively. The optimized extract exhibits significant potential for direct incorporation into various products, including sports drinks, fruit juices, and other nutritional beverages. This extract also has the potential to be a great resource for the design and development of innovative medications, as it provides substantial amounts of GABA and polyphenols. Additionally, it is desirable to further quantify individual phenolic compounds and explore encapsulation technologies, such as encapsulation by freeze drying, for bioactive compounds including GABA and TPC. These approaches would extend the storage time and broaden the application prospects in the food and pharmaceutical industries, enhancing the versatility and stability of these beneficial compounds.
5. ACKNOWLEDGMENTS
The authors would like to express their sincere gratitude to Nong Lam University, Ho Chi Minh City, Vietnam, for providing financial support for the research project CS-CB21-CNHH-01.
6. AUTHOR CONTRIBUTIONS
Anh Thuy Vu, Tuyen Chan Kha, and Huan Tai Phan contributed to experimental design. Anh Thuy Vu carried out the data analysis, manuscript preparation and data interpretation. All authors read and approved the final manuscript.
7. CONFLICTS OF INTEREST
The authors report no financial or any other conflicts of interest in this work.
8. ETHICAL APPROVALS
This study does not involve experiments on animals or human subjects.
9. DATA AVAILABILITY
The authors possess all available data and will furnish it upon request.
10. PUBLISHER’S NOTE
This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
REFERENCES
1. Huang X, Cai W, Xu B. Kinetic changes of nutrients and antioxidant capacities of germinated soybean (Glycine max L.) and mung bean (Vigna radiata L.) with germination time. Food Chem 2014;143:268-76. [CrossRef]
2. Sattar DE, Ali TM, Hasnain A. Effect of different techniques on germination efficacy and antioxidant capacity of indigenous legumes of Pakistan. J Basic Appl Sci 2015;11:348-53. [CrossRef]
3. Gan RY, Lui WY, Wu K, Chan CL, Dai SH, Sui ZQ, et al. Bioactive compounds and bioactivities of germinated edible seeds and sprouts:An updated review. Trends Food Sci Technol 2017;59:1-14. [CrossRef]
4. Hussain I, Burhanddin M. Optimization of germination conditions for germinated mung bean flour by response surface methodology. Afr J Food Sci Technol 2011;2:232-9.
5. Ramos-Ruiz R, Poirot E, Flores-Mosquera M. GABA, a non-protein amino acid ubiquitous in food matrices. Cogent Food Agric 2018;4:1534323. [CrossRef]
6. Nikmaram N, Dar BN, Roohinejad S, Koubaa M, Barba FJ, Greiner R, et al. Recent advances in g-aminobutyric acid (GABA) properties in pulses:An overview. J Sci Food Agric 2017;97:2681-9. [CrossRef]
7. Jannoey P, Niamsup H, Lumyong S, Tajima S, Nomura M, Chairote G. g-aminobutyric acid (GABA) accumulations in rice during germination. Chiang Mai J Sci 2010;37:124-33.
8. Sarasa SB, Mahendran R, Muthusamy G, Thankappan B, Selta DR, Angayarkanni J. A brief review on the non-protein amino acid, gamma-amino butyric acid (GABA):Its production and role in microbes. Curr Microbiol 2020;77:534-44. [CrossRef]
9. Yamuangmorn S, Dell B, Prom-u-thai C. Anthocyanin and phenolic acid profiles in purple, red and non-pigmented rice during germination. Chiang Mai Univ J Nat Sci 2020;19:865-78. [CrossRef]
10. Kanatt SR, Arjun K, Sharma A. Antioxidant and antimicrobial activity of legume hulls. Food Res Int 2011;44:3182-7. [CrossRef]
11. Azmir J, Zaidul IS, Rahman MM, Sharif KM, Mohamed A, Sahena F, et al. Techniques for extraction of bioactive compounds from plant materials:A review. J Food Eng 2013;117:426-36. [CrossRef]
12. Avinash MG, Zameer F, Gopal S. The propensity of selected Indian plant extracts for polyphenolics, antioxidant, and inhibition of Pseudomonas aeruginosa biofilms through Type-3 secretion system. J Appl Biol Biotechnol 2022;10:87-96. [CrossRef]
13. Jha AK, Sit N. Extraction of bioactive compounds from plant materials using combination of various novel methods:A review. Trends Food Sci Technol 2022;119:579-91. [CrossRef]
14. Zhang QW, Lin LG, Ye WC. Techniques for extraction and isolation of natural products:A comprehensive review. Chin Med 2018;13:20. [CrossRef]
15. Kaur A, Singh S, Singh RS, Schwarz WH, Puri M. Hydrolysis of citrus peel naringin by recombinant a-L-rhamnosidase from Clostridium stercorarium. J Chem Technol Biotechnol 2010;85:1419-22. [CrossRef]
16. Puri M, Sharma D, Barrow CJ. Enzyme-assisted extraction of bioactives from plants. Trends Biotechnol 2012;30:37-44. [CrossRef]
17. Nag S, Sit N. Optimization of ultrasound assisted enzymatic extraction of polyphenols from pomegranate peels based on phytochemical content and antioxidant property. J Food Meas Charact 2018;12:1734-43. [CrossRef]
18. Sbroggio MF, Montilha MS, de Figueiredo VR, Georgetti SR, Kurozawa LE. Influence of the degree of hydrolysis and type of enzyme on antioxidant activity of okara protein hydrolysates. Food Sci Technol 2016;36:375-81. [CrossRef]
19. Gardossi L, Poulsen PB, Ballesteros A, Hult K, Svedas VK, Vasi?-Racki D, et al. Guidelines for reporting of biocatalytic reactions. Trends Biotechnol 2010;28:171-80. [CrossRef]
20. Jiang L, Wang W, Wen P, Shen M, Li H, Ren Y, et al. Two water-soluble polysaccharides from mung bean skin:Physicochemical characterization, antioxidant and antibacterial activities. Food Hydrocoll 2020;100:105412. [CrossRef]
21. Xie J, Du M, Shen M, Wu T, Lin L. Physico-chemical properties, antioxidant activities and angiotensin-I converting enzyme inhibitory of protein hydrolysates from mung bean (Vigna radiate). Food Chem 2019;270:243-50. [CrossRef]
22. Nadar SS, Rao P, Rathod VK. Enzyme assisted extraction of biomolecules as an approach to novel extraction technology:A review. Food Res Int 2018;108:309-30. [CrossRef]
23. Vu AT, Kha TC, Phan HT. Effects of soaking and germination conditions on gamma-aminobutyric acid and total phenolic content in geminated mung bean. J Agric Dev 2019;18:112-8. [CrossRef]
24. Vu AT, Kha TC, Phan HT. The changes in gamma-aminobutyric acid and polyphenols in mung beans (Vigna radiata L.) during germination. IOP Conf Ser Earth Environ Sci 2023;1155:012024. [CrossRef]
25. Watchararparpaiboon W, Laohakunjit N, Kerdchoechuen O. An improved process for high quality and nutrition of brown rice production. Food Sci Technol Int 2010;16:147-58. [CrossRef]
26. Z?otek U, Mikulska S, Nagajek M, ?wieca M. The effect of different solvents and number of extraction steps on the polyphenol content and antioxidant capacity of basil leaves (Ocimum basilicum L.) extracts. Saudi J Biol Sci 2016;23:628-33. [CrossRef]
27. Sridhar A, Ponnuchamy M, Kumar PS, Kapoor A, Vo DN, Prabhakar S. Techniques and modeling of polyphenol extraction from food:A review. Environ Chem Lett 2021;19:3409-43. [CrossRef]
28. Wu L, Jiang A, Jing Y, Zheng Y, Yan Y. Antioxidant properties of protein hydrolysate from Douchi by membrane ultrafiltration. Int J Food Prop 2017;20:997-1006. [CrossRef]
29. Hau EH, Teh SS, Yeo SK, Mah SH. Physicochemical and functional properties of Alcalase-extracted protein hydrolysate from oil palm leaves. J Sci Food Agric 2022;102:233-40. [CrossRef]
30. Madia VN, De Vita D, Ialongo D, Tudino V, De Leo A, Scipione L, et al. Recent advances in recovery of lycopene from tomato waste:A potent antioxidant with endless benefits. Molecules 2021;26:4495. [CrossRef]
31. Sánchez-Camargo AD, Montero L, Stiger-Pouvreau V, Tanniou A, Cifuentes A, Herrero M, et al. Considerations on the use of enzyme-assisted extraction in combination with pressurized liquids to recover bioactive compounds from algae. Food Chem 2016;192:67-74. [CrossRef]
32. Parada MI, González HD. Enzyme-assisted aqueous extraction processes. In:Water Extraction of Bioactive Compounds. Netherlands:Elsevier;2017. 333-68. [CrossRef]
33. Fu XQ, Ma N, Sun WP, Dang YY. Microwave and enzyme co-assisted aqueous two-phase extraction of polyphenol and lutein from marigold (Tagetes erecta L.) flower. Ind Crop Prod 2018;123:296-302. [CrossRef]
34. Kha TC, Nguyen MH, Roach PD, Stathopoulos CE. Microencapsulation of Gac oil:Optimisation of spray drying conditions using response surface methodology. Powder Technol 2014;264:298-309. [CrossRef]
35. Tirado-Kulieva VA, Sánchez-Chero M, YarlequéMV, Aguilar GF, Carrión-Barco G, Cruz AG, et al. An overview on the use of response surface methodology to model and optimize extraction processes in the food industry. Curr Res Nutr Food Sci 2021;9:745-54. [CrossRef]
36. Amulya PR, Ul Islam R. Optimization of enzyme-assisted extraction of anthocyanins from eggplant (Solanum melongena L.) peel. Food Chem X 2023;18:100643. [CrossRef]
37. Qadir R, Anwar F, Naseem K, Tahir MH, Alhumade H. Enzyme-assisted extraction of phenolics from Capparis spinosa fruit:Modeling and optimization of the process by RSM and ANN. ACS Omega 2022;7:33031-8. [CrossRef]
38. Su CH, Pham TT, Cheng HH. Aqueous enzymatic extraction of rosmarinic acid from Salvia officinalis:Optimisation using response surface methodology. Phytochem Anal 2020;31:575-82. [CrossRef]